This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
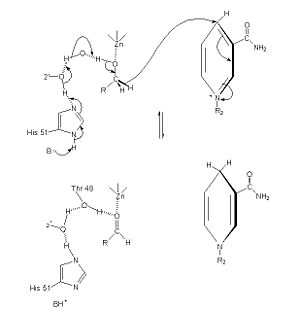
Alcohol dehydrogenase
From Proteopedia
(Difference between revisions)
| Line 62: | Line 62: | ||
{{Clear}} | {{Clear}} | ||
| + | |||
| + | == 3D Structures of alcohol dehydrogenase== | ||
| + | [[Alcohol dehydrogenase 3D structures]] | ||
| + | |||
</StructureSection> | </StructureSection> | ||
| Line 92: | Line 96: | ||
**[[2xaa]] – RrADH I + NAD + alcohol<br /> | **[[2xaa]] – RrADH I + NAD + alcohol<br /> | ||
**[[6ffx]] – RrADH I + Zn + NAD<br /> | **[[6ffx]] – RrADH I + Zn + NAD<br /> | ||
| - | **[[6ffz]], [[5o8h]], [[5o8q]], [[5o9d]], [[5o9f]] – RrADH I (mutant) + Zn + NAD<br /> | + | **[[6ffz]], [[5o8h]], [[5o8q]], [[5o9d]], [[5o9f]], [[6fg0]], [[5od3]] – RrADH I (mutant) + Zn + NAD<br /> |
**[[3fx4]] – pADH I + NADP + inhibitor – pig<br /> | **[[3fx4]] – pADH I + NADP + inhibitor – pig<br /> | ||
**[[4w6z]] – yADH I + Zn + NAD derivative<br /> | **[[4w6z]] – yADH I + Zn + NAD derivative<br /> | ||
| Line 107: | Line 111: | ||
**[[3owo]] – ZmADH II iron-dependent – ''Zymomonas mobilis'' | **[[3owo]] – ZmADH II iron-dependent – ''Zymomonas mobilis'' | ||
| - | *ADH II | + | *ADH II complex |
**[[3ox4]] - ZmADH II iron-dependent + NAD<br /> | **[[3ox4]] - ZmADH II iron-dependent + NAD<br /> | ||
| Line 113: | Line 117: | ||
**[[1e3e]] – mADH II + NADH – mouse<br /> | **[[1e3e]] – mADH II + NADH – mouse<br /> | ||
**[[1e3l]] - mADH II (mutant) + NADH<br /> | **[[1e3l]] - mADH II (mutant) + NADH<br /> | ||
| - | **[[1e3i]] - mADH II + NADH + inhibitor | + | **[[1e3i]] - mADH II + NADH + inhibitor<br /> |
| + | **[[5yln]] – ADH II + Zn – ''Streptococcus pneumoniae''<br /> | ||
*ADH III | *ADH III | ||
| Line 126: | Line 131: | ||
**[[4dla]] – tADH III<br /> | **[[4dla]] – tADH III<br /> | ||
**[[5mln]] – ADH III + NADPH – ''Candida magnoliae''<br /> | **[[5mln]] – ADH III + NADPH – ''Candida magnoliae''<br /> | ||
| + | **[[5yat]] – ADH III + Zn – ''Komagsaataella phaffii''<br /> | ||
*ADH IV | *ADH IV | ||
| Line 154: | Line 160: | ||
**[[5vkr]] - hoADH IV e chain + Zn + ADPR<br /> | **[[5vkr]] - hoADH IV e chain + Zn + ADPR<br /> | ||
**[[5vl0]] - hoADH IV e chain + Zn + formamide derivative <br /> | **[[5vl0]] - hoADH IV e chain + Zn + formamide derivative <br /> | ||
| + | **[[6cy3]], [[6cxx]] - hoADH IV e chain (mutant) + Zn + dephosphoCoA<br /> | ||
**[[5cdu]] - hoADH IV e chain (mutant) + Zn + NAD + pyrazole<br /> | **[[5cdu]] - hoADH IV e chain (mutant) + Zn + NAD + pyrazole<br /> | ||
| Line 207: | Line 214: | ||
**[[4l0q]] - AtADH III (mutant) + NAD + Zn <br /> | **[[4l0q]] - AtADH III (mutant) + NAD + Zn <br /> | ||
**[[4cpd]] – TtADH + NAD + Zn<br /> | **[[4cpd]] – TtADH + NAD + Zn<br /> | ||
| + | **[[6n7l]] – ADH + NAD + Zn – ''Elizabethkingia anophelis''<br /> | ||
| + | **[[6c76]] – TthADH + NADP + Fe – ''Thermococcus thioreducens''<br /> | ||
| + | **[[6c7l]] – TthADH + monophosphoadenosine diphosphate <br /> | ||
| + | **[[6c75]] – TthADH + NADP + Fe + monophosphoadenosine diphosphate <br /> | ||
*NADP-dependent ADH | *NADP-dependent ADH | ||
| Line 237: | Line 248: | ||
**[[5yvm]] – bspADH + Mn + NADP derivative – brine sea pool<br /> | **[[5yvm]] – bspADH + Mn + NADP derivative – brine sea pool<br /> | ||
**[[5yvr]], [[5yvs]] – bspADH (mutant) + Mn + NADP <br /> | **[[5yvr]], [[5yvs]] – bspADH (mutant) + Mn + NADP <br /> | ||
| + | **[[5z2x]] – ADH + NADP – ''Kluyveromyces polyspora''<br /> | ||
*R-specific ADH | *R-specific ADH | ||
| - | **[[1nxq]] - LbRADH – ''Lactobacillus brevis''<br /> | + | **[[1nxq]], [[6h07]], [[6h1m]] - LbRADH – ''Lactobacillus brevis''<br /> |
| - | **[[1zk2]], [[1zk3]] - LbRADH (mutant)<br /> | + | **[[1zk2]], [[1zk3]], [[6hlf]] - LbRADH (mutant)<br /> |
**[[1zjy]], [[1zjz]], [[1zk0]], [[1zk1]] – LbRADH (mutant) + NADH + alcohol<br /> | **[[1zjy]], [[1zjz]], [[1zk0]], [[1zk1]] – LbRADH (mutant) + NADH + alcohol<br /> | ||
**[[1zk4]] - LbRADH (mutant) + NADH + acetophenone<br /> | **[[1zk4]] - LbRADH (mutant) + NADH + acetophenone<br /> | ||
Revision as of 08:29, 4 March 2019
| |||||||||||
Additional Resources
For additional information, see: Carbohydrate Metabolism
3D Structures of Alcohol dehydrogenase
Updated on 04-March-2019
References
- ↑ Voet, et. al. Fundamentals of Biochemistry: 3rd Edition. Hoboken: Wiley & Sons, Inc, 2008.
- ↑ Voet, et. al. Fundamentals of Biochemistry: 3rd Edition. Hoboken: Wiley & Sons, Inc, 2008.
- ↑ Protein: Alcohol Dehydrogenase. The College of Saint Benedict and Saint John's University. 1 March 2010 < http://web.archive.org/web/20080307193453/http://www.users.csbsju.edu/~hjakubow/classes/rasmolchime/99ch331proj/alcoholdehydro/index.htm>
- ↑ Protein: Alcohol Dehydrogenase. The College of Saint Benedict and Saint John's University. 1 March 2010 < http://web.archive.org/web/20080307193453/http://www.users.csbsju.edu/~hjakubow/classes/rasmolchime/99ch331proj/alcoholdehydro/index.htm>
- ↑ Voet, et. al. Fundamentals of Biochemistry: 3rd Edition. Hoboken: Wiley & Sons, Inc, 2008.
- ↑ Dickinson FM, Monger GP. A study of the kinetics and mechanism of yeast alcohol dehydrogenase with a variety of substrates. Biochem J. 1973 Feb;131(2):261-70. PMID:4352908
- ↑ Dickinson FM, Monger GP. A study of the kinetics and mechanism of yeast alcohol dehydrogenase with a variety of substrates. Biochem J. 1973 Feb;131(2):261-70. PMID:4352908
- ↑ Bille V, Remacle J. Simple-kinetic descriptions of alcohol dehydrogenase after immobilization on tresyl-chloride-activated agarose. Eur J Biochem. 1986 Oct 15;160(2):343-8. PMID:3769934
- ↑ Dickinson FM, Monger GP. A study of the kinetics and mechanism of yeast alcohol dehydrogenase with a variety of substrates. Biochem J. 1973 Feb;131(2):261-70. PMID:4352908
- ↑ Blomstrand R, Ostling-Wintzell H, Lof A, McMartin K, Tolf BR, Hedstrom KG. Pyrazoles as inhibitors of alcohol oxidation and as important tools in alcohol research: an approach to therapy against methanol poisoning. Proc Natl Acad Sci U S A. 1979 Jul;76(7):3499-503. PMID:115004
- ↑ Alcohol Dehydrogenase. Worthington Biochemical Corporation . 31 March 2010 < http://http://www.worthington-biochem.com/ADH/default.html>
- ↑ Alcohol Dehydrogenase.Worthington Biochemical Corporation . 31 March 2010 < http://http://www.worthington-biochem.com/ADH/default.html>
- ↑ Goihberg E, Dym O, Tel-Or S, Levin I, Peretz M, Burstein Y. A single proline substitution is critical for the thermostabilization of Clostridium beijerinckii alcohol dehydrogenase. Proteins. 2007 Jan 1;66(1):196-204. PMID:17063493 doi:10.1002/prot.21170
- ↑ Goihberg E, Dym O, Tel-Or S, Shimon L, Frolow F, Peretz M, Burstein Y. Thermal stabilization of the protozoan Entamoeba histolytica alcohol dehydrogenase by a single proline substitution. Proteins. 2008 Feb 7;. PMID:18260103 doi:10.1002/prot.21946
- ↑ Goihberg E, Peretz M, Tel-Or S, Dym O, Shimon L, Frolow F, Burstein Y. Biochemical and Structural Properties of Chimeras Constructed by Exchange of Cofactor-Binding Domains in Alcohol Dehydrogenases from Thermophilic and Mesophilic Microorganisms. Biochemistry. 2010 Feb 9. PMID:20102159 doi:10.1021/bi901730x
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, David Canner, Joel L. Sussman, David Birrer