This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Tilman Schirmer/Sandbox 99
From Proteopedia
(→Cis peptide bonds) |
(→Cis peptide bonds) |
||
| Line 20: | Line 20: | ||
==== Cis peptide bonds ==== | ==== Cis peptide bonds ==== | ||
| - | <scene name='User:Tilman_Schirmer/Sandbox_99/Pro_213_in_cis_conformation/6'>cis-Pro</scene> | ||
| - | <scene name='User:Tilman_Schirmer/Sandbox_99/38/387166/Pro_213_in_cis_conformation/6'>cis-Pro</scene> | ||
| - | <scene name='38/387166/Pro_213_in_cis_conformation/6'> | + | <scene name='38/387166/Pro_213_in_cis_conformation/6'>cis-Pro</scene> |
Revision as of 08:15, 19 September 2019
Contents |
Peptide bond
|
The (highlight in ) formation is a condensation reaction between the carboxyl group of the amino acid i and the amino group of the amino acid i+1.
The peptide bond is a resonance structure between two limiting states. Therefore the N-C bond has a partial double bond character and the atoms/groups Cα, HN, C, C=O are within one plane. Peptide bonds are usually in trans conformation (ω torsion angle= 180°). In cis-conformation, there would be steric hindrance between Cα,i and Cα,i+1.
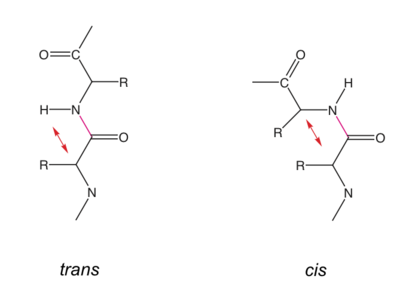
Cis conformation has been observed almost exclusively for peptide bonds preceding a Pro residue (Xaa - Pro), see below.
is defined by the four atoms φ = C - N - Cα - C (in green).
is defined by the four atoms ψ = N - Cα - C - N (in green).
is defined by the four atoms ω = Cα - C - N - Cα (in green).
Cis peptide bonds
The ω torsion angle can adopt a value close to 0° (cis-conformation), when a Pro residue is the following residue (Xaa-Pro peptide bond). In this situation a and a are similarily unfavorable, since there is a steric clash between Cα,i with Cα,i+1 or Cδ,i+1, respectively. Conversely, the carbonyl O of residue i is in tight juxtaposition with Cδ,i+1 or Cα,i+1 (note that latter tight contact occurs in any trans peptide bond).
Torsion angle φ restricted for Pro
Due to the ring closure of the Pro side-chain the torsion around the N - Cα is considerably to a value between -50° to -90°.
References
- Chakrabarti P, Pal D. The interrelationships of side-chain and main-chain conformations in proteins. Prog Biophys Mol Biol. 2001;76(1-2):1-102. PMID:11389934
Next Topic
Secondary structure of proteins http://proteopedia.org/wiki/index.php/User:Tilman_Schirmer/Sandbox_100
