This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1558
From Proteopedia
(Difference between revisions)
| Line 15: | Line 15: | ||
== Broader Implications == | == Broader Implications == | ||
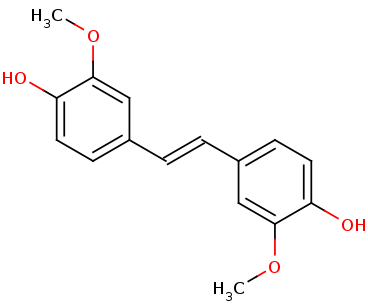
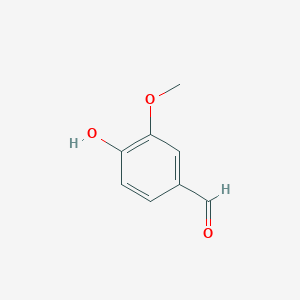
| - | To examine LsdA’s substrate specificity, we heterologously | + | To examine LsdA’s substrate specificity, we heterologously produce the dimeric enzyme with the help of chaperones. When |
| - | tested on several substituted stilbenes, LsdA exhibited the greatest specificity for lignostilbene. These experiments further | + | tested on several substituted stilbenes, LsdA exhibited the greatest specificity for lignostilbene. These experiments further indicate that the substrate’s |
4-hydroxy moiety is required for catalysis and that this moiety | 4-hydroxy moiety is required for catalysis and that this moiety | ||
cannot be replaced with a methoxy group. This expands our | cannot be replaced with a methoxy group. This expands our | ||
Revision as of 01:31, 9 December 2019
| This Sandbox is Reserved from Aug 26 through Dec 12, 2019 for use in the course CHEM 351 Biochemistry taught by Bonnie_Hall at the Grand View University, Des Moines, USA. This reservation includes Sandbox Reserved 1556 through Sandbox Reserved 1575. |
To get started:
More help: Help:Editing |
Lignostilbene-α,β-dioxygenase A (LsdA) Catalyzation
| |||||||||||
References
Kuatsjah, Eugene, et al. “Identification of Functionally Important Residues and Structural Features in a Bacterial Lignostilbene Dioxygenase.” Journal of Biological Chemistry, vol. 294, no. 35, 2019, pp. 12911–12920., doi:10.1074/jbc.ra119.009428.
- ↑ Hanson, R. M., Prilusky, J., Renjian, Z., Nakane, T. and Sussman, J. L. (2013), JSmol and the Next-Generation Web-Based Representation of 3D Molecular Structure as Applied to Proteopedia. Isr. J. Chem., 53:207-216. doi:http://dx.doi.org/10.1002/ijch.201300024
- ↑ Herraez A. Biomolecules in the computer: Jmol to the rescue. Biochem Mol Biol Educ. 2006 Jul;34(4):255-61. doi: 10.1002/bmb.2006.494034042644. PMID:21638687 doi:10.1002/bmb.2006.494034042644