This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Léa Wick/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 2: | Line 2: | ||
== Complex with dibenzofuran-4,6-dicarboxylic acid == | == Complex with dibenzofuran-4,6-dicarboxylic acid == | ||
| + | |||
| + | [[Image: TTR-DDRF1.png | thumb | left | alt=Puzzle globe| TTR in complexe with Dibenzofuran-4,6-dicarboxylic acid (DDBF)| 200 px]] | ||
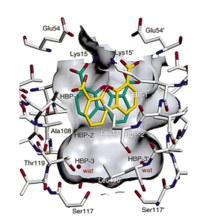
The TTR complexed with dibenzofuran-4,6-dicarboxylic acid has been produced from ''homo sapiens gene'' in ''Escherichia coli'' and it structure determined by X-Ray diffraction ([https://www.rcsb.org/structure/1dvu/1dvu]). | The TTR complexed with dibenzofuran-4,6-dicarboxylic acid has been produced from ''homo sapiens gene'' in ''Escherichia coli'' and it structure determined by X-Ray diffraction ([https://www.rcsb.org/structure/1dvu/1dvu]). | ||
| Line 9: | Line 11: | ||
DDBF is bounded according two symmetric equivalent modes [2]. Indeed, DDBF wears a tricyclic ring system, with 2 hydrogen bond donors and 5 hydrogen bond acceptors, allowing to bound the dimer-dimer interface of the TTR cavity. [Pubchem,1]. Thanks to the complementarity of shape and hydrophobicity, DDBF enters nicely the outer portion of HBPs pockets [2]. Besides, the tricyclic ring system interacts with Lys15, Val17 and Ala108 from two adjacent TTR subunits [2]. Additionally, carboxylates at the position 4 and 6 of DDBF make electrostatic interactions at the entrance of HBP1 and HBP1’ with Lys15 on the ε-NH3+ groups[2]. | DDBF is bounded according two symmetric equivalent modes [2]. Indeed, DDBF wears a tricyclic ring system, with 2 hydrogen bond donors and 5 hydrogen bond acceptors, allowing to bound the dimer-dimer interface of the TTR cavity. [Pubchem,1]. Thanks to the complementarity of shape and hydrophobicity, DDBF enters nicely the outer portion of HBPs pockets [2]. Besides, the tricyclic ring system interacts with Lys15, Val17 and Ala108 from two adjacent TTR subunits [2]. Additionally, carboxylates at the position 4 and 6 of DDBF make electrostatic interactions at the entrance of HBP1 and HBP1’ with Lys15 on the ε-NH3+ groups[2]. | ||
| - | [[Image: EXAMPLE.png | thumb | left | alt=Puzzle globe| TTR in complexe with Dibenzofuran-4,6-dicarboxylic acid (DDBF)| 300 px]] | ||
| - | 1 TTR in complexe with Dibenzofuran-4,6-dicarboxylic acid (DDBF) | ||
TTR in complex with dibenzofuran-4,6-dicarboxylic acid keeps the general apo-structure, with water molecules bind to HBP3 and HBP3’ cavities of TTR [1]. There is not conformational change of Ser117 and Thr119 of TTR, contrary to other inhibitor, such as FLU. | TTR in complex with dibenzofuran-4,6-dicarboxylic acid keeps the general apo-structure, with water molecules bind to HBP3 and HBP3’ cavities of TTR [1]. There is not conformational change of Ser117 and Thr119 of TTR, contrary to other inhibitor, such as FLU. | ||
Consequently, DDBF creates a bridge between two adjacent subunits stabilized by ionic and hydrophobic interactions. | Consequently, DDBF creates a bridge between two adjacent subunits stabilized by ionic and hydrophobic interactions. | ||
Revision as of 12:35, 6 January 2020
| |||||||||||