This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Ivan E. Wang/Sandbox Reserved 895
From Proteopedia
| Line 22: | Line 22: | ||
<StructureSection load='4ryy' size='340' side='right' caption='Figure 3:(R)-emixustat and palmitate bound in the active site of RPE65' scene=''> | <StructureSection load='4ryy' size='340' side='right' caption='Figure 3:(R)-emixustat and palmitate bound in the active site of RPE65' scene=''> | ||
(''R'')-emixustat (ACU-4429) is an investigational small molecule inhibitor of RPE65 first invented by a British-American chemist, Ian L. Scott. Formulated as an hydrochloride salt, (''R'')-emixustat hydrochloride is taken by mouth and functions as a visual cycle modulator (VCM) to reduce toxic retinal byproducts in the canonical visual cycle such as A2E. | (''R'')-emixustat (ACU-4429) is an investigational small molecule inhibitor of RPE65 first invented by a British-American chemist, Ian L. Scott. Formulated as an hydrochloride salt, (''R'')-emixustat hydrochloride is taken by mouth and functions as a visual cycle modulator (VCM) to reduce toxic retinal byproducts in the canonical visual cycle such as A2E. | ||
| + | |||
In 2008, Acucela Inc. partnered with Otsuka Pharmaceutical Company for the continued development of (''R'')-emixustat as a potential inhibitor of RPE65. Currently (''R'')-emixustat is in Phase III clinical trials in the United States for the potential treatment of Stargard's disease, a juvenile form of atrophic (dry) age dependent macular degeneration (AMD). Additionally, (''R'')-emixustat is investigated as potential therapy for diabetic retinopathy and diabetic macular edema. | In 2008, Acucela Inc. partnered with Otsuka Pharmaceutical Company for the continued development of (''R'')-emixustat as a potential inhibitor of RPE65. Currently (''R'')-emixustat is in Phase III clinical trials in the United States for the potential treatment of Stargard's disease, a juvenile form of atrophic (dry) age dependent macular degeneration (AMD). Additionally, (''R'')-emixustat is investigated as potential therapy for diabetic retinopathy and diabetic macular edema. | ||
| + | |||
Historically (''R'')-Emixustat was tested as a possible treatment for dry (atrophic) age-related macular degeneration in Phase IIb/III clinical trials but failed to show clinical outcomes due to significant pharmacokinetic and pharmacodynamic short falls. | Historically (''R'')-Emixustat was tested as a possible treatment for dry (atrophic) age-related macular degeneration in Phase IIb/III clinical trials but failed to show clinical outcomes due to significant pharmacokinetic and pharmacodynamic short falls. | ||
Revision as of 18:24, 25 February 2020
Contents |
Introduction to the RPE65
| |||||||||||
Structure and Activity
Structural Analysis of RPE65
(PLACEHOLDER)
Enzymatic Activity of RPE65
(PLACEHOLDER)
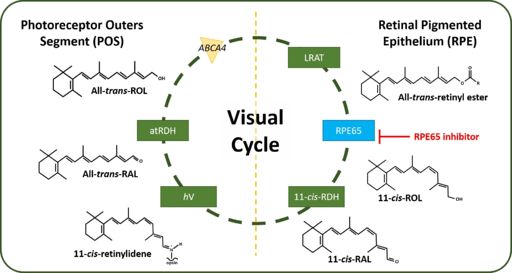
Protein-Ligand Interaction
Endogenous Ligand
(PLACEHOLDER)
Exogenous Ligand
(R)-Emixustat (ACU-4429)
| |||||||||||
(S)-Emixustat
| |||||||||||
R/S Enantiomers Differences
(PLACEHOLDER)
Disease Implications
(PLACEHOLDER)
Medical Relevance
(PLACEHOLDER)
This is a sample scene created with SAT to by Group, and another to make of the protein. You can make your own scenes on SAT starting from scratch or loading and editing one of these sample scenes.
References
- ↑ Shin Y, Moiseyev G, Petrukhin K, Cioffi CL, Muthuraman P, Takahashi Y, Ma JX. A novel RPE65 inhibitor CU239 suppresses visual cycle and prevents retinal degeneration. Biochim Biophys Acta Mol Basis Dis. 2018 Jul;1864(7):2420-2429. doi:, 10.1016/j.bbadis.2018.04.014. Epub 2018 Apr 21. PMID:29684583 doi:http://dx.doi.org/10.1016/j.bbadis.2018.04.014
