This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Daniel Seeman
From Proteopedia
(Difference between revisions)
m |
m |
||
| Line 5: | Line 5: | ||
[[Image:delphiproteins.png|center|thumb|400px|Electrostatic potentials of three proteins (β-lactoglobulin, Bovine serum albumin, and Zn-Insulin) at pH 6. Calculated with DelPhi (a 'Nonlinear Poisson Boltzmann Solver') and displayed using UCSF Chimera. Protein charge anisotropy is a major component of both protein self-association, ''and'' interactions with bio-derived polyelectrolytes.]] | [[Image:delphiproteins.png|center|thumb|400px|Electrostatic potentials of three proteins (β-lactoglobulin, Bovine serum albumin, and Zn-Insulin) at pH 6. Calculated with DelPhi (a 'Nonlinear Poisson Boltzmann Solver') and displayed using UCSF Chimera. Protein charge anisotropy is a major component of both protein self-association, ''and'' interactions with bio-derived polyelectrolytes.]] | ||
| - | + | ||
Current revision
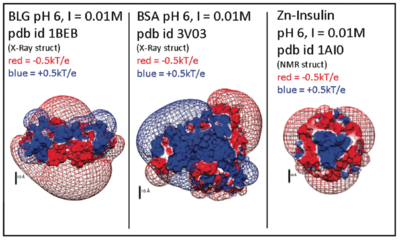
Electrostatic potentials of three proteins (β-lactoglobulin, Bovine serum albumin, and Zn-Insulin) at pH 6. Calculated with DelPhi (a 'Nonlinear Poisson Boltzmann Solver') and displayed using UCSF Chimera. Protein charge anisotropy is a major component of both protein self-association, and interactions with bio-derived polyelectrolytes.
About proteopedia:
- Topic Pages
