This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Dora Bonadio/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 16: | Line 16: | ||
== An attractive drug target == | == An attractive drug target == | ||
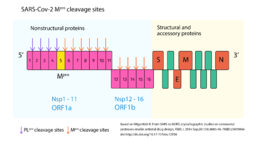
| - | As have been shown, because of its importance for viral replication, inhibiting SARS-CoV-2 Mpro activity could lead to viral replication blockage <ref name="Crystal_structure" /><ref name="ofmpro" />. Moreover, no human proteases has been reported to have a similar cleavage specificity and so, in this aspect, Mpro inhibitors toxic side-effects may be reduced <ref name="sars_mers" />. Therefore, CoV Mpro has been an attractive drug target among coronaviruses <ref name="sars_mers" /> and so it is for COVID-19 <ref name="Crystal_structure" /><ref name="ofmpro" />. Indeed, virtual drug screening, structure-assisted drug design, and high-throughput screening are been used to repurpose approved pharmaceutical drug and drug candidates targeting SARS-CoV-2 Mpro <ref name="ofmpro" /><ref name="elucidation"> Mirza, Muhammad Usman, and Matheus Froeyen. 2020. ‘Structural Elucidation of SARS-CoV-2 Vital Proteins: Computational Methods Reveal Potential Drug Candidates against Main Protease, Nsp12 Polymerase and Nsp13 Helicase’. Journal of Pharmaceutical Analysis, April. Doi:[https://doi.org/10.1016/j.jpha.2020.04.008].</ref>. Furthermore, a study carrying the pharmacokinetic characterization of an optimized Mpro <scene name='84/845941/13b/1'>α-ketoamide inhibitor</scene> provides useful framework for development of this kind of inhibitors toward coronaviruses <ref name="Crystal_structure" />. It was showed that the α-ketoamide inhibitor interacts with the catalytic site of the enzyme through two hydrogen bonding interactions, as can be seen in the complex formed between SARS-CoV-2 Mpro and an α-ketoamide inhibitor <ref name="Crystal_structure" />. | + | As have been shown, because of its importance for viral replication, inhibiting SARS-CoV-2 Mpro activity could lead to viral replication blockage <ref name="Crystal_structure" /><ref name="ofmpro" />. Moreover, no human proteases has been reported to have a similar cleavage specificity and so, in this aspect, Mpro inhibitors toxic side-effects may be reduced <ref name="sars_mers" />. Therefore, CoV Mpro has been an attractive drug target among coronaviruses <ref name="sars_mers" /> and so it is for COVID-19 <ref name="Crystal_structure" /><ref name="ofmpro" />. Indeed, virtual drug screening, structure-assisted drug design, and high-throughput screening are been used to repurpose approved pharmaceutical drug and drug candidates targeting SARS-CoV-2 Mpro <ref name="ofmpro" /><ref name="elucidation"> Mirza, Muhammad Usman, and Matheus Froeyen. 2020. ‘Structural Elucidation of SARS-CoV-2 Vital Proteins: Computational Methods Reveal Potential Drug Candidates against Main Protease, Nsp12 Polymerase and Nsp13 Helicase’. Journal of Pharmaceutical Analysis, April. Doi:[https://doi.org/10.1016/j.jpha.2020.04.008].</ref>. Furthermore, a study carrying the pharmacokinetic characterization of an optimized Mpro <scene name='84/845941/13b/1'>α-ketoamide inhibitor</scene> provides useful framework for development of this kind of inhibitors toward coronaviruses <ref name="Crystal_structure" />. It was showed that the α-ketoamide inhibitor interacts with the catalytic site of the enzyme through two hydrogen bonding interactions, as can be seen in the complex formed between SARS-CoV-2 Mpro and an <scene name='84/845941/13b2/1'>α-ketoamide inhibitor</scene> <ref name="Crystal_structure" />. |
== External Resources == | == External Resources == | ||
Revision as of 08:13, 11 June 2020
SARS-CoV-2 main protease (Mpro)
| |||||||||||