This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Dora Bonadio/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 10: | Line 10: | ||
== Structure == | == Structure == | ||
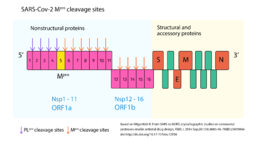
| - | The M<sup>pro</sup> is a protein of approximately 30 kDa <ref name="replication" /><ref name="ofmpro">Jin, Zhenming, Xiaoyu Du, Yechun Xu, Yongqiang Deng, Meiqin Liu, Yao Zhao, Bing Zhang, et al. 2020. ‘Structure of M pro from SARS-CoV-2 and Discovery of Its Inhibitors’. Nature, April, 1–5. https://doi.org/10.1038/s41586-020-2223-y. </ref> consisting of two <scene name='84/845941/Monomer/3'>monomers</scene> containing 306 amino acid residues each <ref name="Crystal_structure" />. This monomers dimerize forming a <scene name='84/845941/Assembly/5'>homodimer</scene> <ref name="Crystal_structure" />. Each chain consists of <scene name='84/845941/Domains/1'>three domains</scene>: I (<scene name='84/845941/Domaini/1'>chymotrypsin-like</scene>; residues 10-99), II (<scene name='84/845941/Domains2/1'>picornavirus 3C protease-like</scene>; residues 100-182), and III (<scene name='84/845941/Domains3/1'>a globular cluster</scene>; residues 198-303). Domains I and II comprise six-stranded antiparallel <scene name='84/845941/B_barrels/1'>β-barrels</scene> and domain III comprises <scene name='84/845941/A_helices/1'>five α-helices</scene> <ref name="Crystal_structure" /><ref name="ofmpro" />. The substrate-binding site is located between domains I and II with the <scene name='84/845941/Catalyticsite/1'>catalytic site</scene> containing the amino acid residues <scene name='84/845941/Cys145_his41/1'>Cys145 and His41</scene> <ref name="Crystal_structure" />. Domain III, in turn, has been shown to be involved in the regulation of M<sup>pro</sup> dimerization, what is necessary for the catalytic activity of this enzyme once it helps to shape the <scene name='84/845941/Substrate_binding_cleft/1'>substrate-binding site</scene> <ref name="Crystal_structure" /><ref name="reveals"> PMID:12093723</ref>. This dimerization regulation is mainly through a <scene name='84/845941/Glu290_arg4/2'>salt-bridge interaction</scene> between Glu290 of one monomer and Arg4 of the other monomer.<ref name="Crystal_structure" /> Moreover, the dimer has a <scene name='84/845941/N_terminal_interaction/1'>contact interface</scene> that is predominantly between domain II of one monomer and the N-terminal residues of other monomer. Indeed, the N-terminal residue <scene name='84/845941/Glu166_ser1/1'>Ser1</scene> of each monomer interacts with Glu166 of the other monomer, helping shape the <scene name='84/845941/Substrate_binding_cleft/1'>substrate-binding site</scene> (notice how Glu166 is a key residue to shape the binding site).<ref name="Crystal_structure" /> | + | The M<sup>pro</sup> is a protein of approximately 30 kDa <ref name="replication" /><ref name="ofmpro">Jin, Zhenming, Xiaoyu Du, Yechun Xu, Yongqiang Deng, Meiqin Liu, Yao Zhao, Bing Zhang, et al. 2020. ‘Structure of M pro from SARS-CoV-2 and Discovery of Its Inhibitors’. Nature, April, 1–5. https://doi.org/10.1038/s41586-020-2223-y. </ref> consisting of two <scene name='84/845941/Monomer/3'>monomers</scene> containing 306 amino acid residues each <ref name="Crystal_structure" />. This monomers dimerize forming a <scene name='84/845941/Assembly/5'>homodimer</scene> <ref name="Crystal_structure" />. Each chain consists of <scene name='84/845941/Domains/1'>three domains</scene>: I (<scene name='84/845941/Domaini/1'>chymotrypsin-like</scene>; residues 10-99), II (<scene name='84/845941/Domains2/1'>picornavirus 3C protease-like</scene>; residues 100-182), and III (<scene name='84/845941/Domains3/1'>a globular cluster</scene>; residues 198-303). Domains I and II comprise six-stranded antiparallel <scene name='84/845941/B_barrels/1'>β-barrels</scene> and domain III comprises <scene name='84/845941/A_helices/1'>five α-helices</scene> <ref name="Crystal_structure" /><ref name="ofmpro" />. The substrate-binding site is located between domains I and II with the <scene name='84/845941/Catalyticsite/1'>catalytic site</scene> containing the amino acid residues <scene name='84/845941/Cys145_his41/1'>Cys145 and His41</scene> <ref name="Crystal_structure" />. Domain III, in turn, has been shown to be involved in the regulation of M<sup>pro</sup> dimerization, what is necessary for the catalytic activity of this enzyme once it helps to shape the <scene name='84/845941/Substrate_binding_cleft/1'>substrate-binding site</scene> <ref name="Crystal_structure" /><ref name="reveals"> PMID:12093723</ref>. This dimerization regulation is mainly through a <scene name='84/845941/Glu290_arg4/2'>salt-bridge interaction</scene> between Glu290 of one monomer and Arg4 of the other monomer.<ref name="Crystal_structure" />. Moreover, the dimer has a <scene name='84/845941/N_terminal_interaction/1'>contact interface</scene> that is predominantly between domain II of one monomer and the N-terminal residues of other monomer. Indeed, the N-terminal residue <scene name='84/845941/Glu166_ser1/1'>Ser1</scene> of each monomer interacts with Glu166 of the other monomer, helping shape the <scene name='84/845941/Substrate_binding_cleft/1'>substrate-binding site</scene> (notice how Glu166 is a key residue to shape the binding site).<ref name="Crystal_structure" /> Therefore, The N-terminal of one monomer interacts with the other monomer by the <scene name='84/845941/Dimerization/1'>Glu166-Ser1 and Glu290-Arg1 interactions</scene> to help dimerization. |
== Structural comparison with SARS-CoV M<sup>pro</sup> == | == Structural comparison with SARS-CoV M<sup>pro</sup> == | ||
Revision as of 00:13, 21 June 2020
SARS-CoV-2 main protease (Mpro)
| |||||||||||