This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Phosphoinositide 3-Kinases
From Proteopedia
(Difference between revisions)
| Line 10: | Line 10: | ||
__TOC__ | __TOC__ | ||
== Function == | == Function == | ||
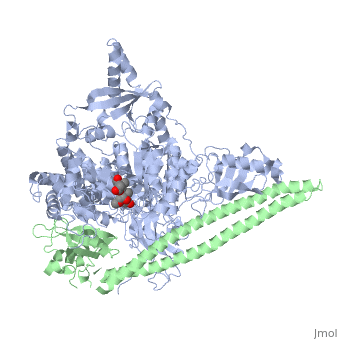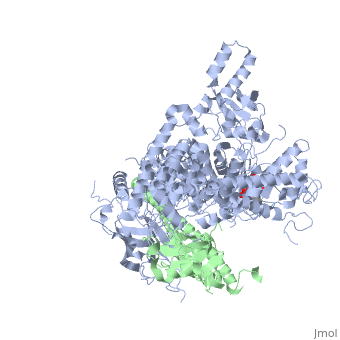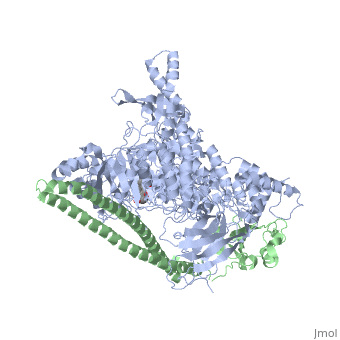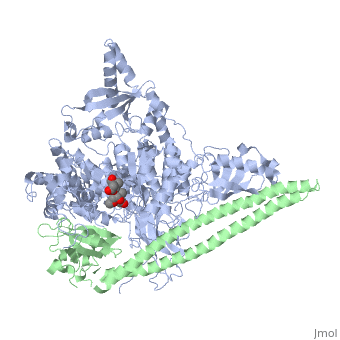
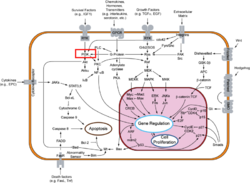
| - | [[Phosphoinositide 3-Kinases]] (PI3K) are a family of ubiquitously distributed lipid kinases, that play a critical role in the regulation of numerous cellular processes including cellular growth and morphology, programmed cell death, cell motility and adhesion, mitogenesis and glucose uptake. <ref name="Driscoll"> PMID: 12151228</ref> PI3K generates important second messengers by catalyzing the transfer of the γ-phosphate group of ATP to the D3 position of phosphoinositides. <ref name="Wymann"> PMID: 9838078</ref> The PI3K preferred substrate is Phosphatidylinositol-4,5-bisphosphate (PIP2), which is converted into phosphatidylinositol-3,4,5-triphosphate (PIP3) upon phosphorylation at the cell membrane. The importance of PI3K is evident in knockout mice studies in which those mice with disruptions of critical PI3K components have significant deficiencies in immune and inflammatory response <ref name="Fubar"> PMID:10972292</ref> sometimes resulting in embryonic death.<ref>PMID:10196176</ref> Aberrations in PIP3 levels, either through activation of PI3ks or through inactivation of lipid phosphatase [[PTEN]], occur frequently in numerous forms of cancer, making PI3K an exciting new target to treat [[Cancer|cancer]] among other human diseases.<ref name="Miled"> PMID: 17626883</ref> For additional details see<br /> | + | [[Phosphoinositide 3-Kinases]] or '''phosphatidylinositol 3-kinase''' (PI3K) are a family of ubiquitously distributed lipid kinases, that play a critical role in the regulation of numerous cellular processes including cellular growth and morphology, programmed cell death, cell motility and adhesion, mitogenesis and glucose uptake. <ref name="Driscoll"> PMID: 12151228</ref> PI3K generates important second messengers by catalyzing the transfer of the γ-phosphate group of ATP to the D3 position of phosphoinositides. <ref name="Wymann"> PMID: 9838078</ref> The PI3K preferred substrate is Phosphatidylinositol-4,5-bisphosphate (PIP2), which is converted into phosphatidylinositol-3,4,5-triphosphate (PIP3) upon phosphorylation at the cell membrane. The importance of PI3K is evident in knockout mice studies in which those mice with disruptions of critical PI3K components have significant deficiencies in immune and inflammatory response <ref name="Fubar"> PMID:10972292</ref> sometimes resulting in embryonic death.<ref>PMID:10196176</ref> Aberrations in PIP3 levels, either through activation of PI3ks or through inactivation of lipid phosphatase [[PTEN]], occur frequently in numerous forms of cancer, making PI3K an exciting new target to treat [[Cancer|cancer]] among other human diseases.<ref name="Miled"> PMID: 17626883</ref> For additional details see<br /> |
* [[PI3K Activation, Inhibition, & Medical Implications]]<br /> | * [[PI3K Activation, Inhibition, & Medical Implications]]<br /> | ||
* [[Human PI3K p110alpha/p85alpha]]<br /> | * [[Human PI3K p110alpha/p85alpha]]<br /> | ||
Revision as of 07:32, 5 October 2020
This page, as it appeared on November 15, 2010, was featured in this article in the journal Biochemistry and Molecular Biology Education.
| |||||||||||
Additional Resources
- See: Cancer For Additional Proteins involved in the disease.
- See: Oncogenes for Additional examples of oncogenes and tumor suppressor genes.
References
- ↑ Djordjevic S, Driscoll PC. Structural insight into substrate specificity and regulatory mechanisms of phosphoinositide 3-kinases. Trends Biochem Sci. 2002 Aug;27(8):426-32. PMID:12151228
- ↑ 2.0 2.1 2.2 Wymann MP, Pirola L. Structure and function of phosphoinositide 3-kinases. Biochim Biophys Acta. 1998 Dec 8;1436(1-2):127-50. PMID:9838078
- ↑ Sasaki T, Irie-Sasaki J, Horie Y, Bachmaier K, Fata JE, Li M, Suzuki A, Bouchard D, Ho A, Redston M, Gallinger S, Khokha R, Mak TW, Hawkins PT, Stephens L, Scherer SW, Tsao M, Penninger JM. Colorectal carcinomas in mice lacking the catalytic subunit of PI(3)Kgamma. Nature. 2000 Aug 24;406(6798):897-902. PMID:10972292 doi:10.1038/35022585
- ↑ Bi L, Okabe I, Bernard DJ, Wynshaw-Boris A, Nussbaum RL. Proliferative defect and embryonic lethality in mice homozygous for a deletion in the p110alpha subunit of phosphoinositide 3-kinase. J Biol Chem. 1999 Apr 16;274(16):10963-8. PMID:10196176
- ↑ Miled N, Yan Y, Hon WC, Perisic O, Zvelebil M, Inbar Y, Schneidman-Duhovny D, Wolfson HJ, Backer JM, Williams RL. Mechanism of two classes of cancer mutations in the phosphoinositide 3-kinase catalytic subunit. Science. 2007 Jul 13;317(5835):239-42. PMID:17626883 doi:317/5835/239
- ↑ Stephens LR, Hughes KT, Irvine RF. Pathway of phosphatidylinositol(3,4,5)-trisphosphate synthesis in activated neutrophils. Nature. 1991 May 2;351(6321):33-9. PMID:1851250 doi:http://dx.doi.org/10.1038/351033a0
- ↑ Hoedemaeker FJ, Siegal G, Roe SM, Driscoll PC, Abrahams JP. Crystal structure of the C-terminal SH2 domain of the p85alpha regulatory subunit of phosphoinositide 3-kinase: an SH2 domain mimicking its own substrate. J Mol Biol. 1999 Oct 1;292(4):763-70. PMID:10525402 doi:http://dx.doi.org/10.1006/jmbi.1999.3111
- ↑ Harris SJ, Foster JG, Ward SG. PI3K isoforms as drug targets in inflammatory diseases: lessons from pharmacological and genetic strategies. Curr Opin Investig Drugs. 2009 Nov;10(11):1151-62. PMID:19876783
Proteopedia Page Contributors and Editors (what is this?)
David Canner, Michal Harel, Joel L. Sussman, Jaime Prilusky, Hannah Campbell, Alexander Berchansky, Angel Herraez