This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Proline utilization A
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
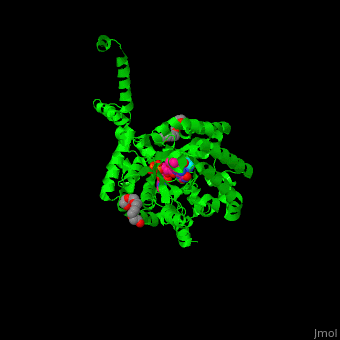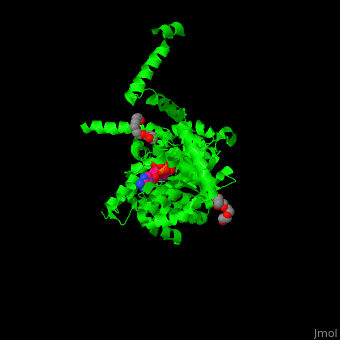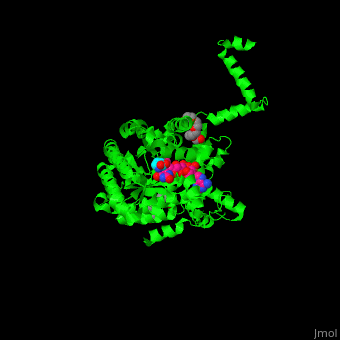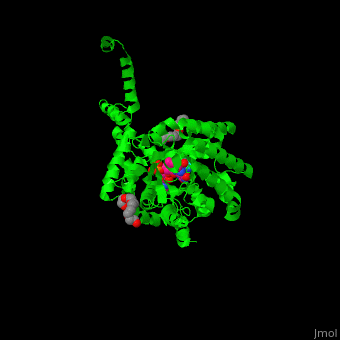
<StructureSection load='' size='350' side='right' caption='E. coli PutA proline dehydrogenase domain with cofactor FAD complex with hydroxyproline (PDB code [[3e2q]])' scene='70/706260/Cv/1'> | <StructureSection load='' size='350' side='right' caption='E. coli PutA proline dehydrogenase domain with cofactor FAD complex with hydroxyproline (PDB code [[3e2q]])' scene='70/706260/Cv/1'> | ||
== Function == | == Function == | ||
| - | '''Proline utilization A (PutA)''' is a bifunctional flavoprotein which acts as a transcriptional repressor of the put regulon or as a membrane-bound enzyme which catalyzes the oxidation of proline to glutamate. PutA domains include the DNA-binding domain (PRODH), the FAD-dependent proline dehydrogenase domain and the D-pyrroline-5-carboxylate dehydrogenase domain. The binding of proline to the PRODH active site and subsequent reduction of FAD causes a conformation change in PutA and enhance its membrane affinity. As a membrane-bound protein PutA switches from its repressor activity to its enzymatic role<ref>PMID:19994913</ref>. | + | '''Proline utilization A (PutA)''' or '''bifunctional protein PutA''' is a bifunctional flavoprotein which acts as a transcriptional repressor of the put regulon or as a membrane-bound enzyme which catalyzes the oxidation of proline to glutamate. PutA domains include the DNA-binding domain (PRODH), the FAD-dependent proline dehydrogenase domain and the D-pyrroline-5-carboxylate dehydrogenase domain. The binding of proline to the PRODH active site and subsequent reduction of FAD causes a conformation change in PutA and enhance its membrane affinity. As a membrane-bound protein PutA switches from its repressor activity to its enzymatic role<ref>PMID:19994913</ref>. |
== Structural highlights == | == Structural highlights == | ||
| Line 11: | Line 11: | ||
Updated on {{REVISIONDAY2}}-{{MONTHNAME|{{REVISIONMONTH}}}}-{{REVISIONYEAR}} | Updated on {{REVISIONDAY2}}-{{MONTHNAME|{{REVISIONMONTH}}}}-{{REVISIONYEAR}} | ||
{{#tree:id=OrganizedByTopic|openlevels=0| | {{#tree:id=OrganizedByTopic|openlevels=0| | ||
| + | |||
*PutA | *PutA | ||
| Line 21: | Line 22: | ||
**[[2gpe]] - EcPutA DNA-binding domain - ''Escherichia coli''<br /> | **[[2gpe]] - EcPutA DNA-binding domain - ''Escherichia coli''<br /> | ||
**[[2jxg]], [[2jxh]] - PpPutA DNA-binding domain - ''Pseudomonas putida'' - NMR<br /> | **[[2jxg]], [[2jxh]] - PpPutA DNA-binding domain - ''Pseudomonas putida'' - NMR<br /> | ||
| - | **[[5ur2]] - PutA + FAD derivative - ''Bdellovibrio bacteriovorus''<br /> | ||
*PutA complex | *PutA complex | ||
| + | **[[6bsn]] – hPutA + FAD derivative + Pro - human<br /> | ||
**[[4nmb]] – GsPutA + FAD + lactate <br /> | **[[4nmb]] – GsPutA + FAD + lactate <br /> | ||
**[[4nmc]] – GsPutA + FAD + zwittergent <br /> | **[[4nmc]] – GsPutA + FAD + zwittergent <br /> | ||
| Line 43: | Line 44: | ||
**[[3haz]] - PutA + FAD + NAD - ''Bradyrhyzobium japonicum''<br /> | **[[3haz]] - PutA + FAD + NAD - ''Bradyrhyzobium japonicum''<br /> | ||
**[[5ux5]] - PutA + FAD + NAD - ''Corynebacterium freiburgense''<br /> | **[[5ux5]] - PutA + FAD + NAD - ''Corynebacterium freiburgense''<br /> | ||
| - | **[[5kf6]], [[5kf7]] - PutA + FAD + NAD + THF - ''Sinorhizobium meliloti''<br /> | + | **[[5kf6]], [[5kf7]], [[6ufp]], [[6vz9]] - PutA + FAD + NAD + THF - ''Sinorhizobium meliloti''<br /> |
**[[2jxi]] - PpPutA DNA-binding domain + DNA<br /> | **[[2jxi]] - PpPutA DNA-binding domain + DNA<br /> | ||
| - | + | **[[5ur2]] - PutA + FAD derivative + inhibitor - ''Bdellovibrio bacteriovorus''<br /> | |
}} | }} | ||
Revision as of 07:52, 7 October 2020
| |||||||||||
3D Structures of proline utilization A
Updated on 07-October-2020
References
- ↑ Srivastava D, Zhu W, Johnson WH, Whitman CP, Becker DF, Tanner JJ. The Structure of the Proline Utilization A Proline Dehydrogenase Domain Inactivated by N-Propargylglycine Provides Insight into Conformational Changes Induced by Substrate Binding and Flavin Reduction (,). Biochemistry. 2009 Dec 29. PMID:19994913 doi:10.1021/bi901717s
- ↑ Ostrander EL, Larson JD, Schuermann JP, Tanner JJ. A Conserved Active Site Tyrosine Residue of Proline Dehydrogenase Helps Enforce the Preference for Proline over Hydroxyproline as the Substrate (dagger) (double dagger). Biochemistry. 2009 Feb 10;48(5):951-9. PMID:19140736 doi:10.1021/bi802094k