This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Estrogen receptor
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
__TOC__ | __TOC__ | ||
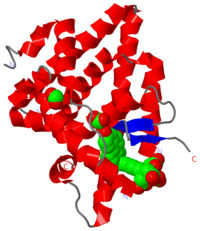
==Estrogen receptors== | ==Estrogen receptors== | ||
| - | (ER) are activated by the hormone estrogen (EST). | + | (ER) are activated by the hormone estrogen (EST). It is a [[Nuclear receptors|nuclear receptor]]. The activated ER binds DNA and regulates the activity of many genes. There are 2 forms of ER: α and β and ER dimers can be of αα, ββ and αβ. ER is composed of 5 domains: N terminal A/B domain can transactivate transcription without binding estrogen; C domain (DBD) binds to Estrogen response elements of DNA; D domain is a hinge region; E domain is ligand binding (LBD) as well as binding the coactivator and corepressor proteins and transactivates gene transcription.<br /> |
Revision as of 16:40, 25 January 2021
| |||||||||||
For more details see
Hormone
Ivan Koutsopatriy estrogen receptor
C-di-GMP receptors with PilZ domain
For more details on ERβ see Student Project 10 for UMass Chemistry 423 Spring 2015
For more details on ER-Tamoxifen complex see Tamoxifen.
Reference
- ↑ Li MJ, Greenblatt HM, Dym O, Albeck S, Pais A, Gunanathan C, Milstein D, Degani H, Sussman JL. Structure of estradiol metal chelate and estrogen receptor complex: The basis for designing a new class of selective estrogen receptor modulators. J Med Chem. 2011 Apr 7. PMID:21473635 doi:10.1021/jm200192y