Testgp
From Proteopedia
(Difference between revisions)
| Line 25: | Line 25: | ||
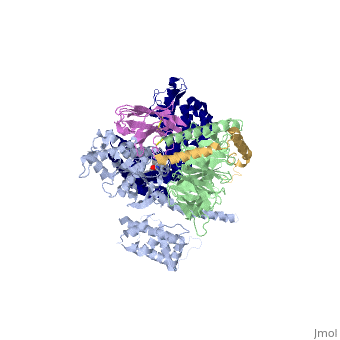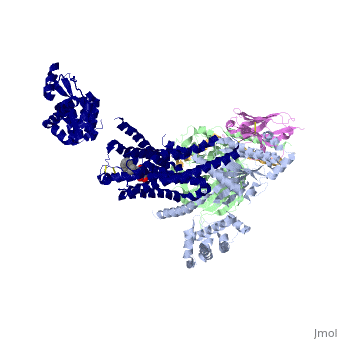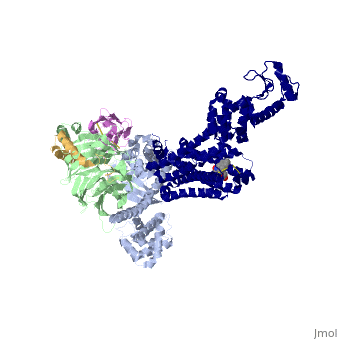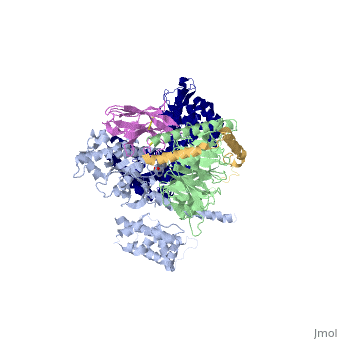
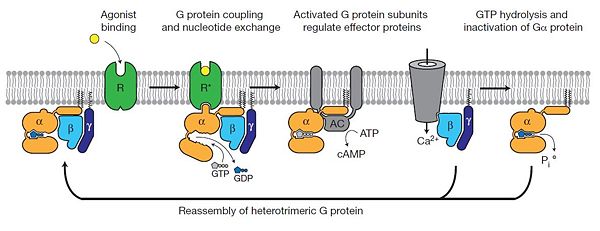
The figure shows the G Protein cycle<ref>doi:10.1038/nature10361</ref> - an extracellular agonist binding to the β2AR leads to <scene name='70/701430/Receptor_morphing_animation/2'>conformational rearrangements</scene> of the cytoplasmic ends of transmembrane segments that enable the Gs heterotrimer to bind the receptor. GDP is released from the α subunit upon formation of β2AR–Gs complex. The GTP binds to the nucleotide-free α subunit resulting in dissociation of the α and βγ subunits from the receptor. The subunits regulate their respective effector proteins adenylyl cyclase (AC) and Ca2+ channels. The Gs heterotrimer reassembles from α and βγ subunits following hydrolysis of GTP to GDP in the α subunit. | The figure shows the G Protein cycle<ref>doi:10.1038/nature10361</ref> - an extracellular agonist binding to the β2AR leads to <scene name='70/701430/Receptor_morphing_animation/2'>conformational rearrangements</scene> of the cytoplasmic ends of transmembrane segments that enable the Gs heterotrimer to bind the receptor. GDP is released from the α subunit upon formation of β2AR–Gs complex. The GTP binds to the nucleotide-free α subunit resulting in dissociation of the α and βγ subunits from the receptor. The subunits regulate their respective effector proteins adenylyl cyclase (AC) and Ca2+ channels. The Gs heterotrimer reassembles from α and βγ subunits following hydrolysis of GTP to GDP in the α subunit. | ||
| + | {{Template:Button Toggle Animation2}} | ||
| + | |||
| + | == G-Protein variability == | ||
| + | The Gαs subunit consists of two domains, the <scene name='70/701430/Alpharas/2'>Ras domain (GαsRas)</scene> and the <scene name='70/701430/Alphahelical/2'>α-helical domain (GαsAH)</scene>. A previous structure of a GTPγS bound (i.e. active, "turned on") Gαs protein (PDB ID: [http://www.rcsb.org/pdb/explore/explore.do?structureId=1AZT 1AZT]) showed that both domains are involved in nucleotide binding, as the nucleotide-binding pocket of the Gαs subunit is formed by the interface between GαsRas and GαsAH<ref>doi:10.1126/science.278.5345.1943</ref>. It was also previously known that the GsαAH domain has a variable position relative to the GsαRas domain between this GTP bound (active) state and the nucleotide free state<ref>DOI:10.1126/science.8266082</ref><ref>doi:10.1073/pnas.1105810108</ref><ref>doi:10.1073/pnas.1113645108</ref><ref>doi:10.1038/nature10488</ref>. However, the β2AR–Gs complex structure of the receptor attached to the empty (no guanosine phosphate attached) G protein enabled comparing it to the active (GTP bound) structure and by that showing <scene name='70/701430/Gamorph/2'>how large this displacement is</scene> - this is probably the most surprising observation arising from the β2AR–Gs complex. | ||
| + | |||
{{Template:Button Toggle Animation2}} | {{Template:Button Toggle Animation2}} | ||
Revision as of 15:13, 25 February 2021
| |||||||||||