This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Madison Unger/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 17: | Line 17: | ||
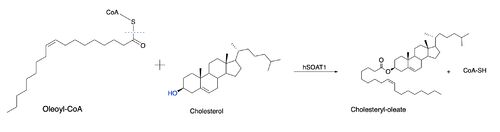
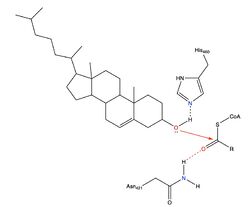
However, Qian et al.<ref>PMID:32433614</ref> proposed a mechanism involving the important residues His460 and Asn421. In this mechanism, His460 acts as a general base to deprotonate the hydroxyl group on cholesterol, activating it as a [http://en.wikipedia.org/wiki/Nucleophile nucleophile]. Then, Asn421 possibly forms a hydrogen bond with oleoyl-CoA to stabilize the reaction. This mechanism is shown in Figure 3. | However, Qian et al.<ref>PMID:32433614</ref> proposed a mechanism involving the important residues His460 and Asn421. In this mechanism, His460 acts as a general base to deprotonate the hydroxyl group on cholesterol, activating it as a [http://en.wikipedia.org/wiki/Nucleophile nucleophile]. Then, Asn421 possibly forms a hydrogen bond with oleoyl-CoA to stabilize the reaction. This mechanism is shown in Figure 3. | ||
===Active Site=== | ===Active Site=== | ||
| + | A <scene name='87/877505/H460_labeled/1'>naturally occurring substrate</scene> can be found in the active site of ACAT and replaced by cholesterol for synthesis of cholesteryl ester. | ||
===Allosteric Binding Pocket=== | ===Allosteric Binding Pocket=== | ||
ACATs are enzymes that can be allosterically activated by sterol molecules like cholesterol. There are two proposed binding sites for cholesterol, with each binding site being distinctively different. One site is the substrate active site mentioned above and the other site is the allosteric binding site. The allosteric binding site has the ability to direct feedback regulation over the concentration of cholesterol in the endoplasmic reticulum. | ACATs are enzymes that can be allosterically activated by sterol molecules like cholesterol. There are two proposed binding sites for cholesterol, with each binding site being distinctively different. One site is the substrate active site mentioned above and the other site is the allosteric binding site. The allosteric binding site has the ability to direct feedback regulation over the concentration of cholesterol in the endoplasmic reticulum. | ||
Revision as of 20:15, 29 March 2021
ACAT/SOAT
| |||||||||||
References
- ↑ Guan C, Niu Y, Chen SC, Kang Y, Wu JX, Nishi K, Chang CCY, Chang TY, Luo T, Chen L. Structural insights into the inhibition mechanism of human sterol O-acyltransferase 1 by a competitive inhibitor. Nat Commun. 2020 May 18;11(1):2478. doi: 10.1038/s41467-020-16288-4. PMID:32424158 doi:http://dx.doi.org/10.1038/s41467-020-16288-4
- ↑ Qian H, Zhao X, Yan R, Yao X, Gao S, Sun X, Du X, Yang H, Wong CCL, Yan N. Structural basis for catalysis and substrate specificity of human ACAT1. Nature. 2020 May;581(7808):333-338. doi: 10.1038/s41586-020-2290-0. Epub 2020 May, 13. PMID:32433614 doi:http://dx.doi.org/10.1038/s41586-020-2290-0
- ↑ Rogers MA, Liu J, Song BL, Li BL, Chang CC, Chang TY. Acyl-CoA:cholesterol acyltransferases (ACATs/SOATs): Enzymes with multiple sterols as substrates and as activators. J Steroid Biochem Mol Biol. 2015 Jul;151:102-7. doi: 10.1016/j.jsbmb.2014.09.008., Epub 2014 Sep 12. PMID:25218443 doi:http://dx.doi.org/10.1016/j.jsbmb.2014.09.008
Student Contributors
- Leah Goehring
- Gabby Smith
- Anna Campbell