We apologize for Proteopedia being slow to respond. For the past two years, a new implementation of Proteopedia has been being built. Soon, it will replace this 18-year old system. All existing content will be moved to the new system at a date that will be announced here.
1jch
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
<StructureSection load='1jch' size='340' side='right'caption='[[1jch]], [[Resolution|resolution]] 3.02Å' scene=''> | <StructureSection load='1jch' size='340' side='right'caption='[[1jch]], [[Resolution|resolution]] 3.02Å' scene=''> | ||
== Structural highlights == | == Structural highlights == | ||
| - | <table><tr><td colspan='2'>[[1jch]] is a 4 chain structure with sequence from [ | + | <table><tr><td colspan='2'>[[1jch]] is a 4 chain structure with sequence from [https://en.wikipedia.org/wiki/Ecow3 Ecow3]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1JCH OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=1JCH FirstGlance]. <br> |
| - | </td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat"><scene name='pdbligand=CIT:CITRIC+ACID'>CIT</scene>, <scene name='pdbligand=GOL:GLYCEROL'>GOL</scene></td></tr> | + | </td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=CIT:CITRIC+ACID'>CIT</scene>, <scene name='pdbligand=GOL:GLYCEROL'>GOL</scene></td></tr> |
| - | <tr id='related'><td class="sblockLbl"><b>[[Related_structure|Related:]]</b></td><td class="sblockDat">[[3eip|3eip]], [[1e44|1e44]]</td></tr> | + | <tr id='related'><td class="sblockLbl"><b>[[Related_structure|Related:]]</b></td><td class="sblockDat"><div style='overflow: auto; max-height: 3em;'>[[3eip|3eip]], [[1e44|1e44]]</div></td></tr> |
| - | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[ | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=1jch FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=1jch OCA], [https://pdbe.org/1jch PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=1jch RCSB], [https://www.ebi.ac.uk/pdbsum/1jch PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=1jch ProSAT]</span></td></tr> |
</table> | </table> | ||
== Function == | == Function == | ||
| - | [[ | + | [[https://www.uniprot.org/uniprot/CEA3_ECOLX CEA3_ECOLX]] Inactivates ribosomes by hydrolyzing 16S RNA in 30S ribosomes at a specific site. Colicins are polypeptide toxins produced by and active against E.coli and closely related bacteria. [[https://www.uniprot.org/uniprot/IMM3_ECOLX IMM3_ECOLX]] This protein inhibits the 16S RNA hydrolyzing activity of colicin E3 by binding with high affinity to the C-terminal catalytic domain of E3. This protein is able to protect a cell, which harbors the plasmid ColE3 against colicin E3. |
== Evolutionary Conservation == | == Evolutionary Conservation == | ||
[[Image:Consurf_key_small.gif|200px|right]] | [[Image:Consurf_key_small.gif|200px|right]] | ||
| Line 33: | Line 33: | ||
*[[Colicin 3D structures|Colicin 3D structures]] | *[[Colicin 3D structures|Colicin 3D structures]] | ||
*[[Colicin immunity protein 3D structures|Colicin immunity protein 3D structures]] | *[[Colicin immunity protein 3D structures|Colicin immunity protein 3D structures]] | ||
| - | *[[Im3|Im3]] | ||
== References == | == References == | ||
<references/> | <references/> | ||
Revision as of 07:18, 14 April 2021
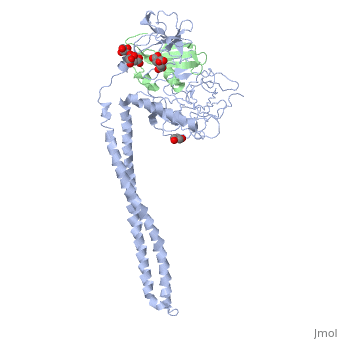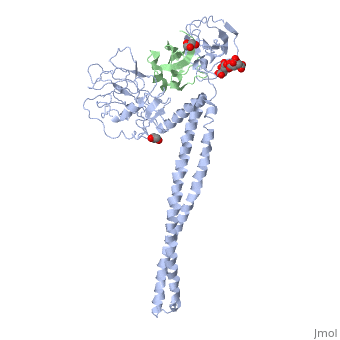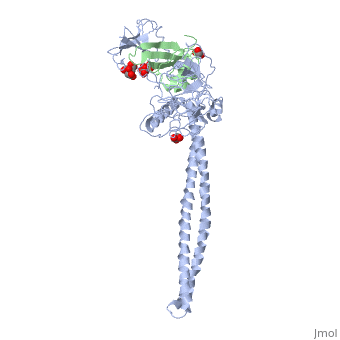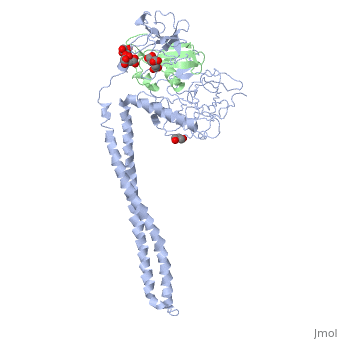
Crystal Structure of Colicin E3 in Complex with its Immunity Protein
| |||||||||||
Categories: Ecow3 | Large Structures | Jakes, K | Li, C | Shoham, M | Soelaiman, S | Wu, N | Hydrolase | Ribosome inhibitor | The receptor-binding domain is a coiled coil | The rnase domain is a six-stranded antiparallel beta-sheet. the immunity protein is a four-stranded antiparallel beta sheet flanked by 3 helices on one side of the sheet | Translocation domain is a beta-jellyroll