This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Megan Fleshman/Sandbox1
From Proteopedia
(Difference between revisions)
| Line 6: | Line 6: | ||
Cholesterol esters were found in arterial lesions in 1910, but the first ACAT activity was discovered in the mid 1900's. This led to the inhibition of ACAT as being looked at as a possible strategy of preventing or treating atherosclerosis. Between 1980-1995, the interest in ACAT inhibitors grew, but some of the compounds looked at exhibited toxicity. As they were looking into the function of the ACAT1 gene, ACAT2 was discovered. In 1993, an ACAT gene was successfully cloned. This discovery led to more studies with ACAT and atherosclerosis. Some of these studies used mice and showed cellular toxicity. ACAT inhibition is still being looked into as a strategy for treatment or prevention of atherosclerosis and related diseases. | Cholesterol esters were found in arterial lesions in 1910, but the first ACAT activity was discovered in the mid 1900's. This led to the inhibition of ACAT as being looked at as a possible strategy of preventing or treating atherosclerosis. Between 1980-1995, the interest in ACAT inhibitors grew, but some of the compounds looked at exhibited toxicity. As they were looking into the function of the ACAT1 gene, ACAT2 was discovered. In 1993, an ACAT gene was successfully cloned. This discovery led to more studies with ACAT and atherosclerosis. Some of these studies used mice and showed cellular toxicity. ACAT inhibition is still being looked into as a strategy for treatment or prevention of atherosclerosis and related diseases. | ||
<ref name=”Farese Jr.”>PMID: 16857957</ref> | <ref name=”Farese Jr.”>PMID: 16857957</ref> | ||
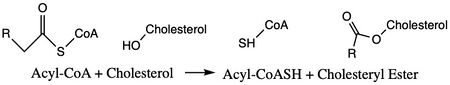
| - | [[Image:simplemechanismforACAT.jpg|450 px|left|thumb|]] | + | [[Image:simplemechanismforACAT.jpg|450 px|left|thumb|Figure 1. Mechanism overview for Acyltransferase of ACAT1]] |
[https://en.wikipedia.org/wiki/Thiolase ACAT] is an important enzyme that catalyzes the esterification of cholesterol to form cholesterol esters, and it belongs to the class of enzymes called acyltransferases. It is also a member of the [https://en.wikipedia.org/wiki/MBOAT MBOAT] family because it is key in lipid metabolism. This enzyme is biologically important because it affects the solubility of cholesterol in the cell membrane and promotes accumulation of cholesterol ester in the cytoplasm as fat droplets. Accumulation of cholesterol ester as these lipid droplets is a main characteristic of macrophage foaming, which can lead to atherosclerotic diseases <ref name=”Qian”>PMID:32433614</ref>. | [https://en.wikipedia.org/wiki/Thiolase ACAT] is an important enzyme that catalyzes the esterification of cholesterol to form cholesterol esters, and it belongs to the class of enzymes called acyltransferases. It is also a member of the [https://en.wikipedia.org/wiki/MBOAT MBOAT] family because it is key in lipid metabolism. This enzyme is biologically important because it affects the solubility of cholesterol in the cell membrane and promotes accumulation of cholesterol ester in the cytoplasm as fat droplets. Accumulation of cholesterol ester as these lipid droplets is a main characteristic of macrophage foaming, which can lead to atherosclerotic diseases <ref name=”Qian”>PMID:32433614</ref>. | ||
| Line 32: | Line 32: | ||
==Mechanism== | ==Mechanism== | ||
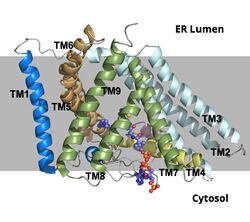
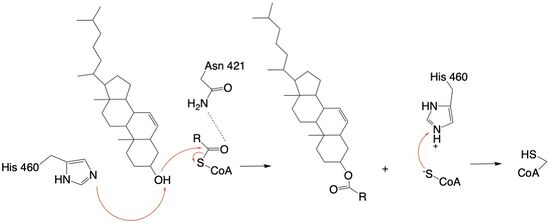
The mechanism of the [[http://en.wikipedia.org/wiki/Acyltransferase#:~:text=Acyltransferase%20is%20a%20type%20of,%2Dalcohol%20O%2Dfatty%2Dacyltransferase acyltransferace]]reaction occurs in the catalytic site one of the monomers in the dimer of ACAT1. The T tunnel and and C tunnel converge to the same space to allow the proper orientation of the Acyl CoA and the incoming cholesterol from the transmembrane. The Acyl CoA is oriented in a way to allow the His460 to act as a base catalyst to begin the reaction by deprotonation of the cholesterol which allows it to attack the carbonyl carbon which breaks the sulfur carbonyl bond (figure 2). The Acyl CoA is held in place by the <scene name='87/877605/Catalytic_residues/4'>oxyanion hold</scene> of Asn 421. This mechanism produces Acyl-CoASH and cholesteryl ester. The Acyl-CcASH leaves through the C tunnel to the cytosol. | The mechanism of the [[http://en.wikipedia.org/wiki/Acyltransferase#:~:text=Acyltransferase%20is%20a%20type%20of,%2Dalcohol%20O%2Dfatty%2Dacyltransferase acyltransferace]]reaction occurs in the catalytic site one of the monomers in the dimer of ACAT1. The T tunnel and and C tunnel converge to the same space to allow the proper orientation of the Acyl CoA and the incoming cholesterol from the transmembrane. The Acyl CoA is oriented in a way to allow the His460 to act as a base catalyst to begin the reaction by deprotonation of the cholesterol which allows it to attack the carbonyl carbon which breaks the sulfur carbonyl bond (figure 2). The Acyl CoA is held in place by the <scene name='87/877605/Catalytic_residues/4'>oxyanion hold</scene> of Asn 421. This mechanism produces Acyl-CoASH and cholesteryl ester. The Acyl-CcASH leaves through the C tunnel to the cytosol. | ||
| - | [[Image:concise mechanism.jpg|550px|left|thumb|Figure | + | [[Image:concise mechanism.jpg|550px|left|thumb|Figure 4: Acyltransferase mechanism of ACAT1 with conserved MBOAT family catalytic residues.]] |
==Inhibitor== | ==Inhibitor== | ||
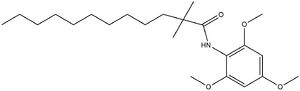
| - | The <scene name='87/877626/Overlay/10'>overlay</scene> illustrates the competitive inhibition of Acetyl-CoA and the inhibitor CI-976. Structurally, Acetyl-CoA and CI-976 are both largely hydrophobic, each with long hydrophobic tails and aromatic heads. As evident in this image, the hydrophobic tail of CI-976, mimics that of Acetyl-CoA. This allows for the inhibitor to be recognized by ACAT and to bind tightly in the active site pocket, blocking Acetyl-CoA from binding, thus rendering ACAT unable to perform its reaction. [[Image: CI-976_chemdraw.jpg|300 px|right|thumb|Figure | + | The <scene name='87/877626/Overlay/10'>overlay</scene> illustrates the competitive inhibition of Acetyl-CoA and the inhibitor CI-976. Structurally, Acetyl-CoA and CI-976 are both largely hydrophobic, each with long hydrophobic tails and aromatic heads. As evident in this image, the hydrophobic tail of CI-976, mimics that of Acetyl-CoA. This allows for the inhibitor to be recognized by ACAT and to bind tightly in the active site pocket, blocking Acetyl-CoA from binding, thus rendering ACAT unable to perform its reaction. [[Image: CI-976_chemdraw.jpg|300 px|right|thumb|Figure 5. CI-976 Inhibitor]] |
Revision as of 00:23, 26 April 2021
Acyl-Coenzyme Cholesterol Acetyltransferase (ACAT)
| |||||||||||
References
ACAT article [8] SOAT Article [9]
- ↑ Farese RV Jr. The nine lives of ACAT inhibitors. Arterioscler Thromb Vasc Biol. 2006 Aug;26(8):1684-6. doi:, 10.1161/01.ATV.0000227511.35456.90. PMID:16857957 doi:http://dx.doi.org/10.1161/01.ATV.0000227511.35456.90
- ↑ Qian H, Zhao X, Yan R, Yao X, Gao S, Sun X, Du X, Yang H, Wong CCL, Yan N. Structural basis for catalysis and substrate specificity of human ACAT1. Nature. 2020 May;581(7808):333-338. doi: 10.1038/s41586-020-2290-0. Epub 2020 May, 13. PMID:32433614 doi:http://dx.doi.org/10.1038/s41586-020-2290-0
- ↑ 3.0 3.1 3.2 3.3 3.4 Chang TY, Chang CC, Bryleva E, Rogers MA, Murphy SR. Neuronal cholesterol esterification by ACAT1 in Alzheimer's disease. IUBMB Life. 2010 Apr;62(4):261-7. doi: 10.1002/iub.305. PMID:20101629 doi:http://dx.doi.org/10.1002/iub.305
- ↑ 4.0 4.1 4.2 4.3 4.4 Shibuya Y, Chang CC, Chang TY. ACAT1/SOAT1 as a therapeutic target for Alzheimer's disease. Future Med Chem. 2015;7(18):2451-67. doi: 10.4155/fmc.15.161. Epub 2015 Dec 15. PMID:26669800 doi:http://dx.doi.org/10.4155/fmc.15.161
- ↑ Ayyagari VN, Wang X, Diaz-Sylvester PL, Groesch K, Brard L. Assessment of acyl-CoA cholesterol acyltransferase (ACAT-1) role in ovarian cancer progression-An in vitro study. PLoS One. 2020 Jan 24;15(1):e0228024. doi: 10.1371/journal.pone.0228024., eCollection 2020. PMID:31978092 doi:http://dx.doi.org/10.1371/journal.pone.0228024
- ↑ Vaziri ND, Liang KH. Acyl-coenzyme A:cholesterol acyltransferase inhibition ameliorates proteinuria, hyperlipidemia, lecithin-cholesterol acyltransferase, SRB-1, and low-denisty lipoprotein receptor deficiencies in nephrotic syndrome. Circulation. 2004 Jul 27;110(4):419-25. doi: 10.1161/01.CIR.0000136023.70841.0F. , Epub 2004 Jul 19. PMID:15262831 doi:http://dx.doi.org/10.1161/01.CIR.0000136023.70841.0F
- ↑ 7.0 7.1 Willner EL, Tow B, Buhman KK, Wilson M, Sanan DA, Rudel LL, Farese RV Jr. Deficiency of acyl CoA:cholesterol acyltransferase 2 prevents atherosclerosis in apolipoprotein E-deficient mice. Proc Natl Acad Sci U S A. 2003 Feb 4;100(3):1262-7. doi: 10.1073/pnas.0336398100., Epub 2003 Jan 21. PMID:12538880 doi:http://dx.doi.org/10.1073/pnas.0336398100
- ↑ Qian H, Zhao X, Yan R, Yao X, Gao S, Sun X, Du X, Yang H, Wong CCL, Yan N. Structural basis for catalysis and substrate specificity of human ACAT1. Nature. 2020 May;581(7808):333-338. doi: 10.1038/s41586-020-2290-0. Epub 2020 May, 13. PMID:32433614 doi:http://dx.doi.org/10.1038/s41586-020-2290-0
- ↑ Guan C, Niu Y, Chen SC, Kang Y, Wu JX, Nishi K, Chang CCY, Chang TY, Luo T, Chen L. Structural insights into the inhibition mechanism of human sterol O-acyltransferase 1 by a competitive inhibitor. Nat Commun. 2020 May 18;11(1):2478. doi: 10.1038/s41467-020-16288-4. PMID:32424158 doi:http://dx.doi.org/10.1038/s41467-020-16288-4
Student Contributors
- Megan Fleshman, Tori Templin, Haylie Moehlenkamp