This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox GGC3
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
<StructureSection loadfiles='4G36''4G37' size='340' side='right' caption='Luciferin-4-monooxygenase. The wild-type luciferase in the adenylate-forming conformation with DLSA (PDB 4G36) and the cross-linked luciferase in the second catalytic conformation with DLSA (PDB 4G37)' scene=''> | <StructureSection loadfiles='4G36''4G37' size='340' side='right' caption='Luciferin-4-monooxygenase. The wild-type luciferase in the adenylate-forming conformation with DLSA (PDB 4G36) and the cross-linked luciferase in the second catalytic conformation with DLSA (PDB 4G37)' scene=''> | ||
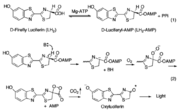
| - | Firefly luciferase, of the common eastern firefly, is responsible for the ability of the firefly to exhibit bioluminescence. The enzyme luciferin-4-monoxygenase, which catalyzes a multistep oxidative decarboxylation of the luciferyl-AMP intermediate (LH<sub>2</sub>-AMP) to produce bioluminescence, is a part of the ANL superfamily named so for the | + | Firefly luciferase, of the common eastern firefly, is responsible for the ability of the firefly to exhibit bioluminescence. The enzyme luciferin-4-monoxygenase, which catalyzes a multistep oxidative decarboxylation of the luciferyl-AMP intermediate (LH<sub>2</sub>-AMP) to produce bioluminescence, is a part of the ANL superfamily named so for the '''a'''cyl-CoA syntheses, the adenylation domains of the modular '''n'''on-ribosomal peptide synthetases (NRPs), and '''l'''uciferase. |
| - | More green links just for fun :~) <scene name='75/752266/Adenylate-forming/2'>adenylate-forming</scene> and <scene name='75/752266/Second_catalytic_conformation/2'>second catalytic conformation</scene> | + | More green links just for fun :~) <scene name='75/752266/Adenylate-forming/2' target='1'>adenylate-forming</scene> and <scene name='75/752266/Second_catalytic_conformation/2'>second catalytic conformation</scene> |
== Function == | == Function == | ||
[[Image:Common_Eastern_Firefly.jpg|thumb|left|The Common Eastern Firefly in a hand emitting a yellow hue, showing bioluminescence.]] | [[Image:Common_Eastern_Firefly.jpg|thumb|left|The Common Eastern Firefly in a hand emitting a yellow hue, showing bioluminescence.]] | ||
| Line 38: | Line 38: | ||
<Structure load='4G36' size='350' frame='true' align='left' caption='Subject to change because why not' scene='Insert optional scene name here' /> | <Structure load='4G36' size='350' frame='true' align='left' caption='Subject to change because why not' scene='Insert optional scene name here' /> | ||
| - | |||
Revision as of 01:43, 27 April 2021
Firefly Luciferase
Check over subscripts and underlineee and tttaaarrgggetttt to the right plaaccceee please oh and why no titles for the references :(
| |||||||||||
References
- ↑ Sundlov, J. A., Fontaine, D. M., Southworth, T. L., Branchini, B. R., Gulick, A. M. (2012). Crystal Structure of Firefly Luciferase in a Second Catalytic Conformation Supports a Domain Alternation Mechanism. Biochemistry 51(33), 6493-6495. https://doi.org/10.1021/bi300934s
- ↑ Marahiel, M. A., Stachelhaus, T., Mootz, H. D. (1997). Modular Peptide Synthetases Involved in Nonribosmal Peptide Synthesis. Chemical Reviews 97(7), 2651-2674. https://doi.org/10.1021/cr960029e
- ↑ Branchini, B. R., Murtiashaw, M. H., Magyar, R. A., Anderson, S. M. (2000). The Role of Lysine 529, a Conserved Residue of the Acyl-Adenylate-Forming Enzyme Superfamily, in Firefly Luciferase. Biochemistry 39(18), 5433-5440. https://doi.org/10.1021/bi9928804
- ↑ Branchini, B. R., Magyar, R. A., Murtiashaw, M. H., Anderson, S. M., Helgerson, L. C., & Zimmer, M. (1999). Site-directed mutagenesis of firefly luciferase active site amino acids: a proposed model for bioluminescence color. Biochemistry 38(40), 13223–13230. https://doi.org/10.1021/bi991181o