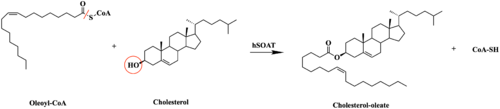
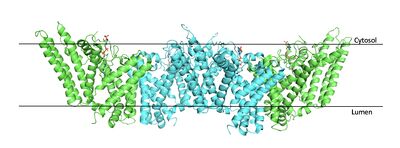
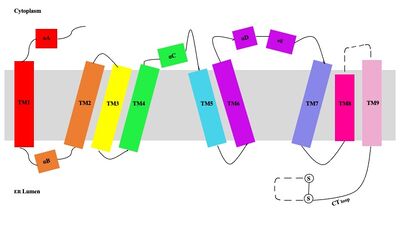
This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Kaitlyn Roberts/Sandbox 2
From Proteopedia
(Difference between revisions)
| Line 12: | Line 12: | ||
=== Tunnel System === | === Tunnel System === | ||
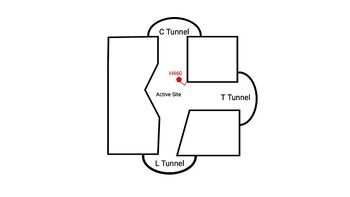
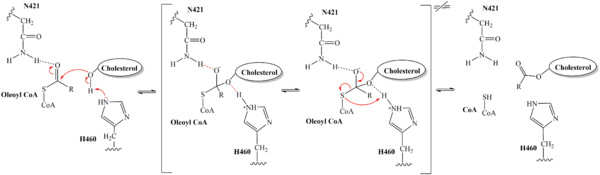
| - | An important structural element of SOAT is the tunnel system through which substrates enter and exit. [[Image:2D_Tunnels.jpg|350 px|right|thumb|'''Figure 4. 2D layout of the proposed SOAT tunnel system.''' The C tunnel opens into the cytosol and the L tunnel opens to the lumen. The T tunnel opens into the membrane, but is not quite oriented at a 90 degree angle as depicted here.]] There are three main tunnels in each monomer: the cytosolic (C) tunnel opens to the cytosol, the transmembrane (T) tunnel opens to the membrane, and the lumenal (L) tunnel opens to the lumen. <ref name="Qian" /> The <scene name='87/877561/Ctunnel_final/1'>C tunnel</scene> is the entrance site for the acyl-CoA substrates, allowing them acess to the active site. Residues <scene name='87/877559/ | + | An important structural element of SOAT is the tunnel system through which substrates enter and exit. [[Image:2D_Tunnels.jpg|350 px|right|thumb|'''Figure 4. 2D layout of the proposed SOAT tunnel system.''' The C tunnel opens into the cytosol and the L tunnel opens to the lumen. The T tunnel opens into the membrane, but is not quite oriented at a 90 degree angle as depicted here.]] There are three main tunnels in each monomer: the cytosolic (C) tunnel opens to the cytosol, the transmembrane (T) tunnel opens to the membrane, and the lumenal (L) tunnel opens to the lumen. <ref name="Qian" /> The <scene name='87/877561/Ctunnel_final/1'>C tunnel</scene> is the entrance site for the acyl-CoA substrates, allowing them acess to the active site. Residues <scene name='87/877559/C_tunnel_and_measurements_fina/1'>N415, Y433, and K445</scene> exhibit hydrogen bonding interactions with polar atoms of coenzyme A to help stabilize the substrate within the binding pocket. Surface representations of SOAT indicate that there are two alpha helices that block the entrance to the C tunnel, therefore a conformational change needs to occur before the substrate can enter the tunnel. The <scene name='87/877561/Ttunnel_final/1'>T tunnel</scene> opens into the membrane and provides access for cholesterol to enter the active site. At the active site, SOAT catalizes the esterification reaction and the products exit through the tunnels. The CoA-SH product exits through the C tunnel and is released back into the cytosol. The cholesteryl ester product exits through either the T tunnel into the membrane or through the <scene name='87/877561/Ltunnelfinal/1'>L tunnel</scene> |
into the lumen of the cell. <ref name="Qian" /> | into the lumen of the cell. <ref name="Qian" /> | ||
| Line 26: | Line 26: | ||
== Inhibitors == | == Inhibitors == | ||
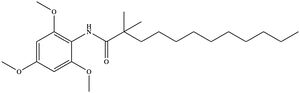
| - | [[Image:InhibitorCI976.jpeg|300 px|right|thumb|'''Figure 6. Structure of the CI-976 inhibitor''']] Depending on concentration and exposure, SOAT activity can be inhibited by CI-976. When exposed, CI-976 locks itself in the <scene name='87/877559/ | + | [[Image:InhibitorCI976.jpeg|300 px|right|thumb|'''Figure 6. Structure of the CI-976 inhibitor''']] Depending on concentration and exposure, SOAT activity can be inhibited by CI-976. When exposed, CI-976 locks itself in the <scene name='87/877559/Inhibitor_in_active_site_final/1'>catalytic center</scene> of the enzyme. The <scene name='87/877559/Residues_and_inhibitor/20'>trimethoxyphenyl head</scene> becomes sandwiched between the catalytic residues H460, W420, and N421. The interaction with these residues, as well as the location of the trimethoxyphenol head, indicate that CI-976 acts as a [https://en.wikipedia.org/wiki/Competitive_inhibition competitive inhibitor] of SOAT by preventing substrates from accessing the catalytic center. Similar to the interactions with the substrates, mutating those key catalytic residues, N421A, H460A, and H460N, result in a smaller effect of the inhibitor on the thermostability of the enzyme. <ref name="Guan" /> |
== Biological Relevance == | == Biological Relevance == | ||
Revision as of 15:20, 27 April 2021
Human Sterol O-acyltransferase
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 Guan C, Niu Y, Chen SC, Kang Y, Wu JX, Nishi K, Chang CCY, Chang TY, Luo T, Chen L. Structural insights into the inhibition mechanism of human sterol O-acyltransferase 1 by a competitive inhibitor. Nat Commun. 2020 May 18;11(1):2478. doi: 10.1038/s41467-020-16288-4. PMID:32424158 doi:http://dx.doi.org/10.1038/s41467-020-16288-4
- ↑ 2.0 2.1 2.2 2.3 Qian H, Zhao X, Yan R, Yao X, Gao S, Sun X, Du X, Yang H, Wong CCL, Yan N. Structural basis for catalysis and substrate specificity of human ACAT1. Nature. 2020 May;581(7808):333-338. doi: 10.1038/s41586-020-2290-0. Epub 2020 May, 13. PMID:32433614 doi:http://dx.doi.org/10.1038/s41586-020-2290-0
- ↑ Das A, Davis MA, Rudel LL. Identification of putative active site residues of ACAT enzymes. J Lipid Res. 2008 Aug;49(8):1770-81. doi: 10.1194/jlr.M800131-JLR200. Epub 2008, May 13. PMID:18480028 doi:http://dx.doi.org/10.1194/jlr.M800131-JLR200
- ↑ Guo ZY, Lin S, Heinen JA, Chang CC, Chang TY. The active site His-460 of human acyl-coenzyme A:cholesterol acyltransferase 1 resides in a hitherto undisclosed transmembrane domain. J Biol Chem. 2005 Nov 11;280(45):37814-26. doi: 10.1074/jbc.M508384200. Epub 2005, Sep 8. PMID:16154994 doi:http://dx.doi.org/10.1074/jbc.M508384200
- ↑ 5.0 5.1 Bhattacharyya R, Kovacs DM. ACAT inhibition and amyloid beta reduction. Biochim Biophys Acta. 2010 Aug;1801(8):960-5. doi: 10.1016/j.bbalip.2010.04.003. , Epub 2010 Apr 14. PMID:20398792 doi:http://dx.doi.org/10.1016/j.bbalip.2010.04.003
- ↑ 6.0 6.1 Huttunen HJ, Kovacs DM. ACAT as a drug target for Alzheimer's disease. Neurodegener Dis. 2008;5(3-4):212-4. doi: 10.1159/000113705. Epub 2008 Mar 6. PMID:18322393 doi:http://dx.doi.org/10.1159/000113705
- ↑ Chang C, Dong R, Miyazaki A, Sakashita N, Zhang Y, Liu J, Guo M, Li BL, Chang TY. Human acyl-CoA:cholesterol acyltransferase (ACAT) and its potential as a target for pharmaceutical intervention against atherosclerosis. Acta Biochim Biophys Sin (Shanghai). 2006 Mar;38(3):151-6. doi:, 10.1111/j.1745-7270.2006.00154.x. PMID:16518538 doi:http://dx.doi.org/10.1111/j.1745-7270.2006.00154.x
- ↑ Ayyagari VN, Wang X, Diaz-Sylvester PL, Groesch K, Brard L. Assessment of acyl-CoA cholesterol acyltransferase (ACAT-1) role in ovarian cancer progression-An in vitro study. PLoS One. 2020 Jan 24;15(1):e0228024. doi: 10.1371/journal.pone.0228024., eCollection 2020. PMID:31978092 doi:http://dx.doi.org/10.1371/journal.pone.0228024
Student Contributors
- Kylie Pfeifer
- Stephanie Pellegrino
- Kaitlyn Roberts