This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Hexokinase
From Proteopedia
| Line 21: | Line 21: | ||
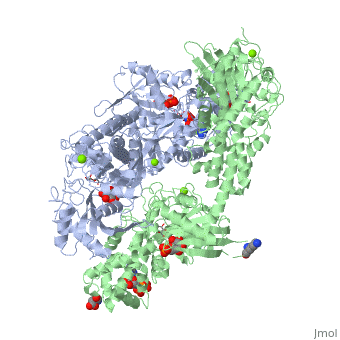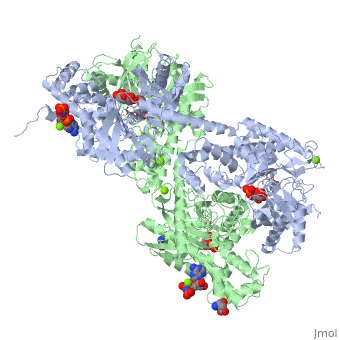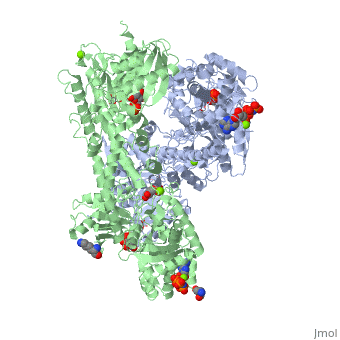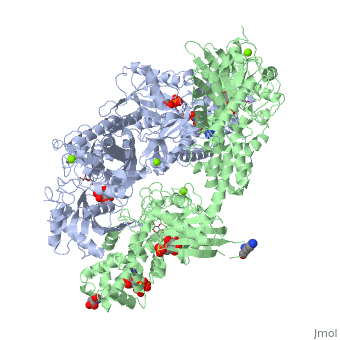
== Conformational change associated with substrate binding == | == Conformational change associated with substrate binding == | ||
| - | When hexokinase binds to glucose (one of its two substrates), it exhibits induced fit. This means that in the open form of the enzyme, the binding site is not fully formed. Upon binding glucose, hexokinase switches into a closed form, excluding aqueous solvent from the substrate. This is illustrated here <scene name='45/452482/Induced_fit/ | + | When hexokinase binds to glucose (one of its two substrates), it exhibits induced fit. This means that in the open form of the enzyme, the binding site is not fully formed. Upon binding glucose, hexokinase switches into a closed form, excluding aqueous solvent from the substrate. This is illustrated here <scene name='45/452482/Induced_fit/2'>comparing the structures of free and glucose-bound hexokinase</scene>. |
<jmol> | <jmol> | ||
| Line 28: | Line 28: | ||
<script>anim off; delay 0.5; model 1</script> | <script>anim off; delay 0.5; model 1</script> | ||
<text>Open</text> | <text>Open</text> | ||
| + | <checked>true</checked> | ||
</item> | </item> | ||
<item> | <item> | ||
<script>anim off; delay 0.5; model 2</script> | <script>anim off; delay 0.5; model 2</script> | ||
<text>Close</text> | <text>Close</text> | ||
| + | <checked>false</checked> | ||
</item> | </item> | ||
<item> | <item> | ||
<script>anim mode palindrome; anim on</script> | <script>anim mode palindrome; anim on</script> | ||
<text>Animate</text> | <text>Animate</text> | ||
| - | <checked> | + | <checked>false</checked> |
</item> | </item> | ||
</jmolRadioGroup> | </jmolRadioGroup> | ||
| Line 43: | Line 45: | ||
It is a bit easier to see the extent and nature of the changes in this <jmol> | It is a bit easier to see the extent and nature of the changes in this <jmol> | ||
<jmolLink> | <jmolLink> | ||
| - | <script> script "/scripts/45/452482/Induced_fit/ | + | <script> script "/scripts/45/452482/Induced_fit/2.spt"; script "/wiki/images/a/a2/Storymorph.spt"; model 2; color background black; |
structures = [{1.1},{1.2}]; | structures = [{1.1},{1.2}]; | ||
domain2 = {82-210}; | domain2 = {82-210}; | ||
Revision as of 18:22, 30 April 2021
| |||||||||||
Additional Resources
For additional information, see: Carbohydrate Metabolism
References
1.↑ Pollard-Knight D, Cornish-Bowden A. Mechanism of liver glucokinase. Mol Cell Biochem. 1982 Apr 30;44(2):71-80. PMID:7048063
2.↑ 2.0 2.1 Kamata K, Mitsuya M, Nishimura T, Eiki J, Nagata Y. Structural basis for allosteric regulation of the monomeric allosteric enzyme human glucokinase. Structure. 2004 Mar;12(3):429-38. PMID:15016359 doi:10.1016/j.str.2004.02.005
3.↑ Postic C, Shiota M, Magnuson MA. Cell-specific roles of glucokinase in glucose homeostasis. Recent Prog Horm Res. 2001;56:195-217. PMID:11237213
4.↑ Zeng C, Aleshin A, Hardie J, Harrison R, Fromm H. ATP-Binding site of Human Brain Hexokinase as Studied by Molecular Modeling and Site-Directed Mutagenesis. Biochem. 1996 Aug 6;35:13157-13164.
5.↑ hammes G, and Kochavi D. Studies of the Enzyme Hexokinase: Kinetic Inhibition by Products. Massachusetts Institute of Technology. 1961 Oct 5.
6.↑ Ralph E, Thomson J, Almaden J, Sun S. Glucose Modulation fo Glucokinase Activation by Small Molecules. Biochem. 2008 Feb 15;47:5028-5036.
7.↑ Pal P, and Miller B. Activating Mutations in the Human Glucokinase Gene Revealed by Genetic Selection. Biochem. 2008 Dec 3;48:814-816.
8.↑ Aleshin A, Malfois M, Liu X, Kim C, Fromm H, Honzatko R, Koch M, Svergun D. Nonaggregating Mutant of Recombinant Human Hexokinase I Exhibits Wild-Type Kinetics and Rod-like Conformations in Solution. Biochem. 1999 Apr 29;38:8359-8366.
Seth Bawel and Kyle_Schroering created this page in Che 361 at Wabash College.
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Karsten Theis, Ann Taylor, Alexander Berchansky