This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Veronika Pelekhov/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
''' SN1 התמרה נוקלאופילית - מנגנון ''' [[en:SN1 reaction]] | ''' SN1 התמרה נוקלאופילית - מנגנון ''' [[en:SN1 reaction]] | ||
| + | == S<sub>N</sub>1-Substitution of Cl<sup>-</sup> and ''tert''-Butanol == | ||
| + | <StructureSection load='' size='340' side='right' caption='' scene='54/542276/Side_view/1'> | ||
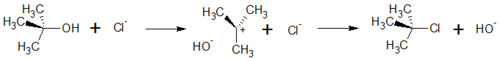
| + | The S<sub>N</sub>1 reaction belongs to the basic reaction in organic chemistry. The number 1 says that it is a monomolecular reaction. This means that in the rate determining step of the reaction, only one of the educts is involved. The kinetic of the reaction therefore follows the reation rate of first order. | ||
| + | |||
| + | In general, substitutions exchange substituents in an organic molecule. One example of an S<sub>N</sub>1 reactions are the exchange of the Hydroxide in <i>tert</i>-Butanol by a Chloride Ion or | ||
| + | |||
| + | In general, SN1 substitution can take place when a stable carbocation can be formed. If not, the reaction follows the SN2 mechanism. | ||
| + | The SN1 <jmol><jmolLink><script>anim mode once; frame range 1 10; delay 0.5; frame play</script><text>reaction starts</text></jmolLink></jmol> with the removement of a hydroxide-ion out of the molecule, in this case tert-Butanol. By this, a positively charged carbocation with a planar geometry is formed. This step is also the rate-determing step because it is the slowest step in this reaction. | ||
| + | In the <jmol><jmolLink><script>anim mode once; frame range 11 20; delay 0.5; frame play</script><text>second step</text></jmolLink></jmol>, the haloanion bound to the carbocation, and a neutral haloalkane is formed. With this step, the hydroxy-substituent is replaced by a halogen-substituent. | ||
| + | |||
| + | [[Image:reaction_scheme_sn1.jpg|500px]] | ||
| + | |||
| + | <jmol> | ||
| + | <jmolButton><script>frame 1</script><text>First</text></jmolButton> | ||
| + | <jmolButton><script>frame prev</script><text>Previous</text></jmolButton> | ||
| + | <jmolButton><script>anim mode once; frame 1; delay 0.5; anim on</script><text>Play</text></jmolButton> | ||
| + | <jmolButton><script>frame next</script><text>Next</text></jmolButton> | ||
| + | </jmol> | ||
| + | |||
| + | <jmol> | ||
| + | <jmolButton><script>frame all</script><text>All frames</text></jmolButton> | ||
| + | <jmolButton><script>animation mode palindrome 0.5 0.2; anim on</script><text>Loop backwards and forward</text></jmolButton> | ||
| + | <jmolButton><script>if(_animating);anim off;else;frame play;endif</script><text>Toggle animation</text></jmolButton> | ||
| + | </jmol> | ||
| + | ===See also=== | ||
| + | [[SN2_reaction|S<sub>N</sub>2 reaction: Substitution of chloride and methanol]] | ||
| + | == References == | ||
| + | This demo was adapted from http://www.chemieunterricht-interaktiv.de/en/animations/sn1_substutition/sn1_substitution_3d.html by Dr. V. Pietzner, part of the ChiLe project | ||
| + | <references/> | ||
| + | </StructureSection> | ||
| + | [[Category: BioMolViz]] | ||
| + | [[Category: Chemical Reactions]] | ||
Revision as of 07:30, 5 July 2021
SN1 התמרה נוקלאופילית - מנגנון
SN1-Substitution of Cl- and tert-Butanol
| |||||||||||