This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Esterification
From Proteopedia
| Line 43: | Line 43: | ||
In the <jmol><jmolLink><script>anim mode once; frame range 11 19; delay 0.5; frame play</script><text>second step</text></jmolLink></jmol>, the yyyy. | In the <jmol><jmolLink><script>anim mode once; frame range 11 19; delay 0.5; frame play</script><text>second step</text></jmolLink></jmol>, the yyyy. | ||
In the <jmol><jmolLink><script>anim mode once; frame range 20 41; delay 0.5; frame play</script><text>third step</text></jmolLink></jmol>, the zzzz. | In the <jmol><jmolLink><script>anim mode once; frame range 20 41; delay 0.5; frame play</script><text>third step</text></jmolLink></jmol>, the zzzz. | ||
| - | In the <jmol><jmolLink><script>anim mode once; frame range 41 | + | In the <jmol><jmolLink><script>anim mode once; frame range 41 50; delay 0.5; frame play</script><text>fourth step</text></jmolLink></jmol>, the aaaa. |
| - | In the <jmol><jmolLink><script>anim mode once; frame range | + | In the <jmol><jmolLink><script>anim mode once; frame range 51 59; delay 0.5; frame play</script><text>fifth step</text></jmolLink></jmol>, the bbbb. |
Revision as of 12:43, 18 July 2021
Contents |
Esterification
Esterification is a chemical reaction of an acid with an alcohol (R'OH) to form an ester (RCOOR').
Usually esterification refers to reaction between an organic (carboxylic) acid (RCOOH) with an alcohol (R'OH) to form an ester (RCOOR') and water and called Fischer esterification.
The chemical reaction for Fischer esterification is given below:
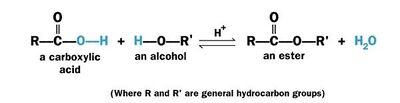
Esterification of fatty acid with ethanol
Esterification of fatty acid with ethanol |
| Drag the structure with the mouse to rotate |
The with protonation of the carbonyl group of the carboxylic acid by the catalyst (H+).
In the , the yyyy.
In the , the zzzz.
In the , the aaaa.
In the , the bbbb.
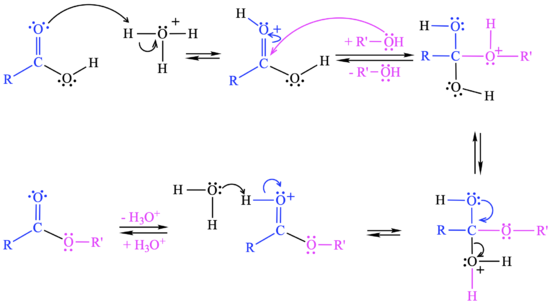
An animated example of this reaction is shown. Please click on the buttons below to animate the reaction with different representations. Use the popup button to enlarge the view and the quality button to turn on anti-aliasing.
The animation was originally done by Prof. Dr. Verena Pietzner; for details, see her web site ChiLe[1]. The implementation into Proteopedia was done by Prof. Jaime Prilusky, Prof. Joel L. Sussman and Veronika Pelekhov.
See also
SN1 reaction: Substitution of Cl− and tert-Butanol
SN2 reaction: substitution of Cl− and methanol
