This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
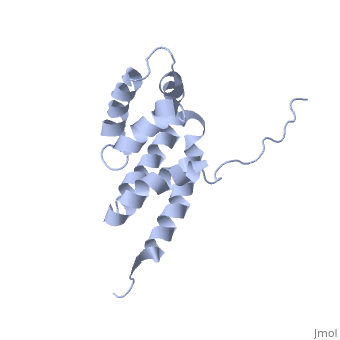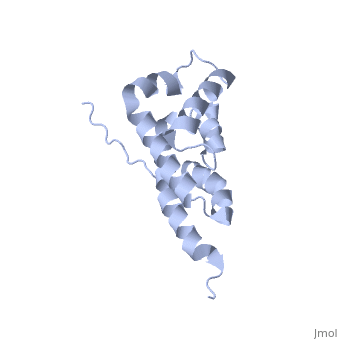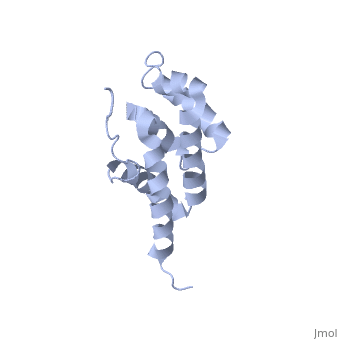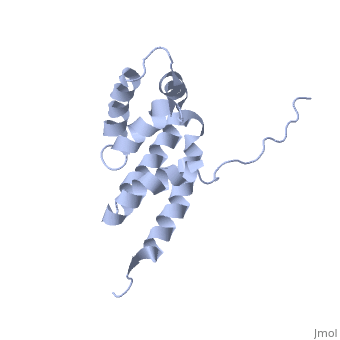
2bo5
From Proteopedia
| Line 3: | Line 3: | ||
<StructureSection load='2bo5' size='340' side='right'caption='[[2bo5]], [[NMR_Ensembles_of_Models | 44 NMR models]]' scene=''> | <StructureSection load='2bo5' size='340' side='right'caption='[[2bo5]], [[NMR_Ensembles_of_Models | 44 NMR models]]' scene=''> | ||
== Structural highlights == | == Structural highlights == | ||
| - | <table><tr><td colspan='2'>[[2bo5]] is a 1 chain structure with sequence from [ | + | <table><tr><td colspan='2'>[[2bo5]] is a 1 chain structure with sequence from [https://en.wikipedia.org/wiki/Bovin Bovin]. Full experimental information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2BO5 OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=2BO5 FirstGlance]. <br> |
| - | </td></tr><tr id='activity'><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[ | + | </td></tr><tr id='activity'><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[https://en.wikipedia.org/wiki/H(+)-transporting_two-sector_ATPase H(+)-transporting two-sector ATPase], with EC number [https://www.brenda-enzymes.info/php/result_flat.php4?ecno=3.6.3.14 3.6.3.14] </span></td></tr> |
| - | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[ | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=2bo5 FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=2bo5 OCA], [https://pdbe.org/2bo5 PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=2bo5 RCSB], [https://www.ebi.ac.uk/pdbsum/2bo5 PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=2bo5 ProSAT]</span></td></tr> |
</table> | </table> | ||
== Function == | == Function == | ||
| - | [[ | + | [[https://www.uniprot.org/uniprot/ATPO_BOVIN ATPO_BOVIN]] Mitochondrial membrane ATP synthase (F(1)F(0) ATP synthase or Complex V) produces ATP from ADP in the presence of a proton gradient across the membrane which is generated by electron transport complexes of the respiratory chain. F-type ATPases consist of two structural domains, F(1) - containing the extramembraneous catalytic core and F(0) - containing the membrane proton channel, linked together by a central stalk and a peripheral stalk. During catalysis, ATP synthesis in the catalytic domain of F(1) is coupled via a rotary mechanism of the central stalk subunits to proton translocation. Part of the complex F(0) domain and the peripheric stalk, which acts as a stator to hold the catalytic alpha(3)beta(3) subcomplex and subunit a/ATP6 static relative to the rotary elements. |
== Evolutionary Conservation == | == Evolutionary Conservation == | ||
[[Image:Consurf_key_small.gif|200px|right]] | [[Image:Consurf_key_small.gif|200px|right]] | ||
Revision as of 14:46, 17 November 2021
Bovine oligomycin sensitivity conferral protein N-terminal domain
| |||||||||||
Categories: Bovin | Large Structures | Carbajo, R J | Kellas, F A | Montgomery, M G | Neuhaus, D | Runswick, M J | Walker, J E | Alpha-subunit | Atp synthase | Beta-subunit | Binding interface | Chemical shift mapping | Chemical shift perturbation | Hydrogen ion transport | Hydrolase | Ion transport | Mitochondrion | Oscp | Peripheral stalk | Protein-protein interaction | Titration | Transit peptide | Transport