This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
2dy5
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
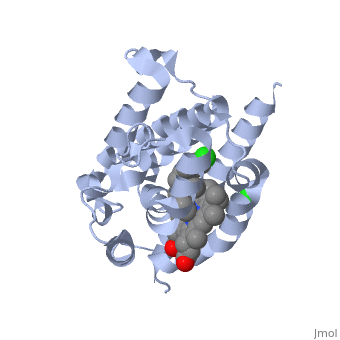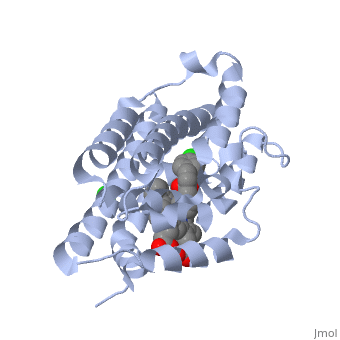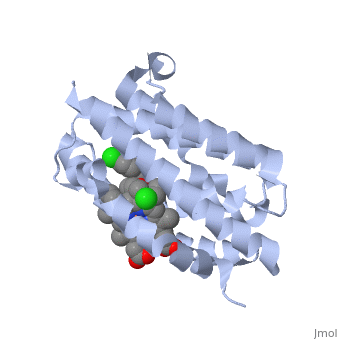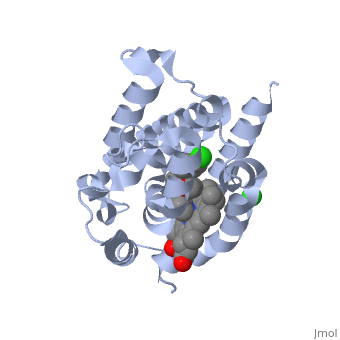
<StructureSection load='2dy5' size='340' side='right'caption='[[2dy5]], [[Resolution|resolution]] 2.70Å' scene=''> | <StructureSection load='2dy5' size='340' side='right'caption='[[2dy5]], [[Resolution|resolution]] 2.70Å' scene=''> | ||
== Structural highlights == | == Structural highlights == | ||
| - | <table><tr><td colspan='2'>[[2dy5]] is a 1 chain structure with sequence from [ | + | <table><tr><td colspan='2'>[[2dy5]] is a 1 chain structure with sequence from [https://en.wikipedia.org/wiki/Buffalo_rat Buffalo rat]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2DY5 OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=2DY5 FirstGlance]. <br> |
| - | </td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat"><scene name='pdbligand=224:1-({2-[2-(4-CHLOROPHENYL)ETHYL]-1,3-DIOXOLAN-2-YL}METHYL)-1H-IMIDAZOLE'>224</scene>, <scene name='pdbligand=CL:CHLORIDE+ION'>CL</scene>, <scene name='pdbligand=HEM:PROTOPORPHYRIN+IX+CONTAINING+FE'>HEM</scene></td></tr> | + | </td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=224:1-({2-[2-(4-CHLOROPHENYL)ETHYL]-1,3-DIOXOLAN-2-YL}METHYL)-1H-IMIDAZOLE'>224</scene>, <scene name='pdbligand=CL:CHLORIDE+ION'>CL</scene>, <scene name='pdbligand=HEM:PROTOPORPHYRIN+IX+CONTAINING+FE'>HEM</scene></td></tr> |
| - | <tr id='activity'><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[ | + | <tr id='activity'><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[https://en.wikipedia.org/wiki/Heme_oxygenase Heme oxygenase], with EC number [https://www.brenda-enzymes.info/php/result_flat.php4?ecno=1.14.99.3 1.14.99.3] </span></td></tr> |
| - | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[ | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=2dy5 FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=2dy5 OCA], [https://pdbe.org/2dy5 PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=2dy5 RCSB], [https://www.ebi.ac.uk/pdbsum/2dy5 PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=2dy5 ProSAT]</span></td></tr> |
</table> | </table> | ||
== Function == | == Function == | ||
| - | [[ | + | [[https://www.uniprot.org/uniprot/HMOX1_RAT HMOX1_RAT]] Heme oxygenase cleaves the heme ring at the alpha methene bridge to form biliverdin. Biliverdin is subsequently converted to bilirubin by biliverdin reductase. Under physiological conditions, the activity of heme oxygenase is highest in the spleen, where senescent erythrocytes are sequestrated and destroyed. |
== Evolutionary Conservation == | == Evolutionary Conservation == | ||
[[Image:Consurf_key_small.gif|200px|right]] | [[Image:Consurf_key_small.gif|200px|right]] | ||
Revision as of 17:18, 15 December 2021
Crystal structure of rat heme oxygenase-1 in complex with heme and 2-[2-(4-chlorophenyl)ethyl]-2-[(1H-imidazol-1-yl)methyl]-1,3-dioxolane
| |||||||||||