This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1660
From Proteopedia
(Difference between revisions)
| Line 37: | Line 37: | ||
:- an <scene name='86/868193/Alpha_chain/7'>alpha chain</scene> (T-cell surface glycoprotein CD1d1) of 287 amino acids <br> | :- an <scene name='86/868193/Alpha_chain/7'>alpha chain</scene> (T-cell surface glycoprotein CD1d1) of 287 amino acids <br> | ||
:- a <scene name='86/868193/Beta-2-microglobulin_chain/6'>beta-2-microglobulin chain</scene> of 99 amino acids. <br> | :- a <scene name='86/868193/Beta-2-microglobulin_chain/6'>beta-2-microglobulin chain</scene> of 99 amino acids. <br> | ||
| - | The alpha chain is made of three domains: alpha 1, alpha 2 and alpha 3. The association of alpha 1 and alpha 2 is composed of two [https://en.wikipedia.org/wiki/Beta_sheet beta-sheets] and a set of 2 [ | + | The alpha chain is made of three domains: alpha 1, alpha 2 and alpha 3. The association of alpha 1 and alpha 2 is composed of two [https://en.wikipedia.org/wiki/Beta_sheet beta-sheets] and a set of 2 [https://proteopedia.org/wiki/index.php/Alpha_helix alpha helixes]. Each beta-sheet contains four antiparallel strands. The ligand binds between the two alpha 1 and 2 helices. The alpha 3 domain is non covalently bound with beta-2-microglobulin domain. |
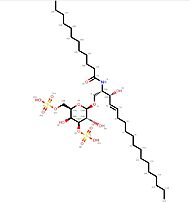
Additionally, there are five <scene name='86/868193/Oligosaccharides/3'>oligosaccharides</scene><ref name="Structure"/> bound to the alpha chain via N-glycosylations, three of which have been clearly identified<ref name="oligo">Sriram, V., Willard, C.A., Liu, J., & Brutkiewicz, R.R.(2008). Importance of N-linked glycosylation in the functional expression of murine CD1d1. Immunology, 123:272–281.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2433293/</ref>. The total molecular weight of the alpha chain is 33 kDa when not associated to any oligosaccharide and 55 kDa when oligosaccharides are associated to the chain.<br> | Additionally, there are five <scene name='86/868193/Oligosaccharides/3'>oligosaccharides</scene><ref name="Structure"/> bound to the alpha chain via N-glycosylations, three of which have been clearly identified<ref name="oligo">Sriram, V., Willard, C.A., Liu, J., & Brutkiewicz, R.R.(2008). Importance of N-linked glycosylation in the functional expression of murine CD1d1. Immunology, 123:272–281.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2433293/</ref>. The total molecular weight of the alpha chain is 33 kDa when not associated to any oligosaccharide and 55 kDa when oligosaccharides are associated to the chain.<br> | ||
CD1d molecules are structurally similar to [[Major Histocompatibility Complex Class I]], but present lipid antigens as opposed to peptides. Thus the cleft where the ligand can bind is different between MHC molecules and CD1d molecules. Indeed, the hydrophobic cleft of CD1d has a narrow opening.The recognition between the protein and its ligand occurs at a specific hydrophobic spot which creates an appropriate environment for the interaction to happen. This <scene name='86/868193/Site/2'>site</scene> is located at the A' and F' pockets in the region of the alpha helices<ref name="site">Schiefner, A.; Fujio, M.; Wu, D.; Wong, C.-H.; Wilson, I. A. Structural Evaluation of Potent NKT-Cell Agonists: Implications for Design of Novel Stimulatory Ligands. J Mol Biol 2009, 394 (1), 71–82. https://doi.org/10.1016/j.jmb.2009.08.061</ref>. | CD1d molecules are structurally similar to [[Major Histocompatibility Complex Class I]], but present lipid antigens as opposed to peptides. Thus the cleft where the ligand can bind is different between MHC molecules and CD1d molecules. Indeed, the hydrophobic cleft of CD1d has a narrow opening.The recognition between the protein and its ligand occurs at a specific hydrophobic spot which creates an appropriate environment for the interaction to happen. This <scene name='86/868193/Site/2'>site</scene> is located at the A' and F' pockets in the region of the alpha helices<ref name="site">Schiefner, A.; Fujio, M.; Wu, D.; Wong, C.-H.; Wilson, I. A. Structural Evaluation of Potent NKT-Cell Agonists: Implications for Design of Novel Stimulatory Ligands. J Mol Biol 2009, 394 (1), 71–82. https://doi.org/10.1016/j.jmb.2009.08.061</ref>. | ||
Revision as of 18:06, 17 January 2022
| This Sandbox is Reserved from 26/11/2020, through 26/11/2021 for use in the course "Structural Biology" taught by Bruno Kieffer at the University of Strasbourg, ESBS. This reservation includes Sandbox Reserved 1643 through Sandbox Reserved 1664. |
To get started:
More help: Help:Editing |
| |||||||||||