This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1713
From Proteopedia
(Difference between revisions)
| Line 7: | Line 7: | ||
== Structure & Function == | == Structure & Function == | ||
===Domains=== | ===Domains=== | ||
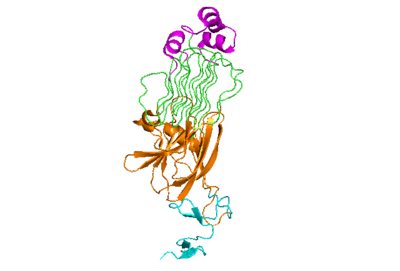
| - | The extracellular portion of ALK has an inactive state, which is its monomerized form, and an active dimerized state with its ligands bound. The monomer is shown to the right in Figure 1, which has many different domains. The growth factor-like domain (EGF) connects the extracellular domains to the transmembrane domain (cyan). The tumor necrosis factor-like domain (TNFL) has a beta-sandwich structure that provides important residues that act as the binding surface for the ligand(orange). The glycine-rich domain (GlyR) contains 14 rare polyglycine helices that are hydrogen-bound to each other (green). The <scene name='90/904318/Glycinerichdomain/1'>hexagonal orientation</scene> of these rare helices create a very rigid structure that is important for ALK function. The polyglycine extension loop (PXL) connects two of these polyglycine helices. | + | The extracellular portion of ALK has an inactive state, which is its monomerized form, and an active dimerized state with its ligands bound. The monomer is shown to the right in Figure 1, which has many different domains. The growth factor-like domain (EGF) connects the extracellular domains to the transmembrane domain (cyan). The tumor necrosis factor-like domain (TNFL) has a beta-sandwich structure that provides important residues that act as the binding surface for the ligand (orange). The glycine-rich domain (GlyR) contains 14 rare polyglycine helices that are hydrogen-bound to each other (green). The <scene name='90/904318/Glycinerichdomain/1'>hexagonal orientation</scene> of these rare helices create a very rigid structure that is important for ALK function. The polyglycine extension loop (PXL) connects two of these polyglycine helices (pink). |
| - | [[Image:Prote_ALK_Monomer_White.png|400 px|right|thumb|Figure 1]] | + | [[Image:Prote_ALK_Monomer_White.png|400 px|right|thumb|Figure 1: The ALK monomer unbound to ALKAL. Cyan: growth factor-like domain (EGF). Orange: tumor necrosis factor-like domain (TNFL). Green: glycine-rich domain (GlyR). Pink: polyglycine extension loop (PXL).]] |
The domains that aren't shown in Figure 1 but are shown in the domain map (Figure 2) that also make up the monomer are the heparin binding domains (HBDs), which are at the N-terminal end of the monomer. Heparin has been found to be a possible activating ligand of ALK. The transmembrane domain (TMH) are the residues of ALK that are located within the membrane. The kinase domain is the intracellular portion of ALK that contains the Tyr residues which are auto-phosphorylated when ALK is activated, initiating a signaling cascade. | The domains that aren't shown in Figure 1 but are shown in the domain map (Figure 2) that also make up the monomer are the heparin binding domains (HBDs), which are at the N-terminal end of the monomer. Heparin has been found to be a possible activating ligand of ALK. The transmembrane domain (TMH) are the residues of ALK that are located within the membrane. The kinase domain is the intracellular portion of ALK that contains the Tyr residues which are auto-phosphorylated when ALK is activated, initiating a signaling cascade. | ||
Revision as of 03:08, 28 March 2022
| This Sandbox is Reserved from February 28 through September 1, 2022 for use in the course CH462 Biochemistry II taught by R. Jeremy Johnson at the Butler University, Indianapolis, USA. This reservation includes Sandbox Reserved 1700 through Sandbox Reserved 1729. |
To get started:
More help: Help:Editing |
Anaplastic Lymphoma Kinase
| |||||||||||
References
- ↑ Reshetnyak AV, Rossi P, Myasnikov AG, Sowaileh M, Mohanty J, Nourse A, Miller DJ, Lax I, Schlessinger J, Kalodimos CG. Mechanism for the activation of the anaplastic lymphoma kinase receptor. Nature. 2021 Dec;600(7887):153-157. doi: 10.1038/s41586-021-04140-8. Epub 2021, Nov 24. PMID:34819673 doi:http://dx.doi.org/10.1038/s41586-021-04140-8