This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1709
From Proteopedia
| Line 7: | Line 7: | ||
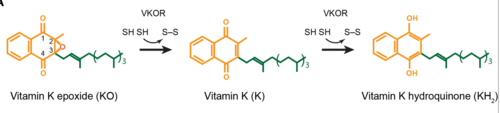
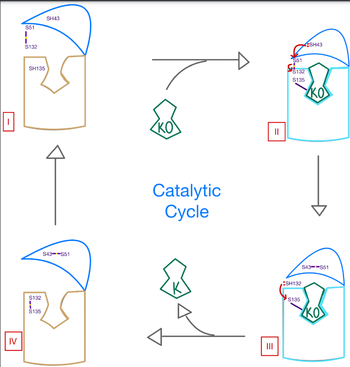
<scene name='90/906893/Vkor_structure/1'>Vitamin K epoxide reductase</scene> (VKOR) is an enzyme that, as its name implies, promotes the reduction of <scene name='90/906893/Vkor_with_ko/1'>vitamin K epoxide</scene> (KO). VKOR is a transmembrane protein spanning the endoplasmic reticulum and composed of [https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2919313/ 4 transmembrane helical proteins]. One of its primary roles is to assist in blood coagulation by regenerating hydroquinone (KH2). KH2 acts as a γ-carboxylase cofactor that drives the γ-carboxylation of several coagulation factors. Structural characterization of VKOR has been difficult, though, due to its in vitro instability. Nonetheless, a near perfect atomic structure has been determined utilization anticoagulant stabilization and VKOR-like [https://pubmed.ncbi.nlm.nih.gov/33154105/ homologs]. | <scene name='90/906893/Vkor_structure/1'>Vitamin K epoxide reductase</scene> (VKOR) is an enzyme that, as its name implies, promotes the reduction of <scene name='90/906893/Vkor_with_ko/1'>vitamin K epoxide</scene> (KO). VKOR is a transmembrane protein spanning the endoplasmic reticulum and composed of [https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2919313/ 4 transmembrane helical proteins]. One of its primary roles is to assist in blood coagulation by regenerating hydroquinone (KH2). KH2 acts as a γ-carboxylase cofactor that drives the γ-carboxylation of several coagulation factors. Structural characterization of VKOR has been difficult, though, due to its in vitro instability. Nonetheless, a near perfect atomic structure has been determined utilization anticoagulant stabilization and VKOR-like [https://pubmed.ncbi.nlm.nih.gov/33154105/ homologs]. | ||
| - | === Function and Biological Role === | ||
[[Image:VKOR_mechanism_2D.png |500 px|right| thumb]] | [[Image:VKOR_mechanism_2D.png |500 px|right| thumb]] | ||
=== Author's Notes === | === Author's Notes === | ||
Revision as of 01:26, 29 March 2022
| |||||||||||
References
1. Elshaikh, A. O., Shah, L., Joy Mathew, C., Lee, R., Jose, M. T., & Cancarevic, I. "Influence of Vitamin K on Bone Mineral Density and Osteoporosis" (2020) Cureus, 12(10), e10816. [1]
2. Li, Weikai et al. “Structure of a bacterial homologue of vitamin K epoxide reductase.” Nature vol. 463,7280 (2010): 507-12. doi:10.1038/nature08720.
3. Liu S, Li S, Shen G, Sukumar N, Krezel AM, Li W. Structural basis of antagonizing the vitamin K catalytic cycle for anticoagulation. Science. 2021 Jan 1;371(6524):eabc5667. doi: 10.1126/science.abc5667. Epub 2020 Nov 5. PMID: 33154105; PMCID: PMC7946407.
4. “Warfarin.” Wikipedia, Wikimedia Foundation, 10 Feb. 2022, https://en.wikipedia.org/wiki/Warfarin.
5. Yang W., et. al. “VKORC1 Haplotypes Are Associated With Arterial Vascular Diseases (Stroke, Coronary Heart Disease, and Aortic Dissection)” (2006) Circulation. ;113:1615–1621 [2]
- ↑ Hanson, R. M., Prilusky, J., Renjian, Z., Nakane, T. and Sussman, J. L. (2013), JSmol and the Next-Generation Web-Based Representation of 3D Molecular Structure as Applied to Proteopedia. Isr. J. Chem., 53:207-216. doi:http://dx.doi.org/10.1002/ijch.201300024
- ↑ Herraez A. Biomolecules in the computer: Jmol to the rescue. Biochem Mol Biol Educ. 2006 Jul;34(4):255-61. doi: 10.1002/bmb.2006.494034042644. PMID:21638687 doi:10.1002/bmb.2006.494034042644