Sandbox Reserved 1716
From Proteopedia
(Difference between revisions)
| Line 46: | Line 46: | ||
===Cap Domain=== | ===Cap Domain=== | ||
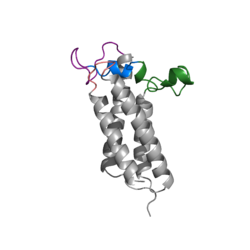
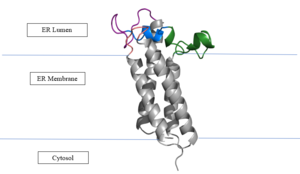
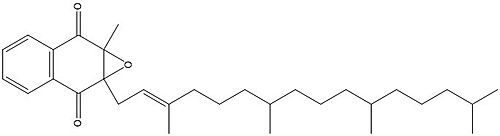
| - | The cap domain of Vitamin K Epoxide Reductase plays an intricate role in its function. When Vitamin K Epoxide (or a similar substrate) binds in the hydrophobic pocket of VKOR, the cap domain undergoes a conformational change that will allow for specific cysteine residues to be able to open the epoxide ring and to recreate Vitamin K Quinone. The catalytic cycle begins in an open fully oxidized conformation. <scene name='90/904321/Openvkor/3'>Open Oxidized Conformation</scene> This conformation has slightly different parts. These include the Anchor (green), the cap region (blue), 3-4 Loop (pink), and luminal helix (yellow). When Vitamin K Epoxide binds, the entire cap domain undergoes a slight conformation change, but the luminal helix has a larger change. The luminal helix (yellow) bends forward where specific cysteines on this region are in proximity to other important cysteines. The luminal helix is then referred to as the beta hairpin (purple). <scene name='90/904321/Vkoclosed/ | + | The cap domain of Vitamin K Epoxide Reductase plays an intricate role in its function. When Vitamin K Epoxide (or a similar substrate) binds in the hydrophobic pocket of VKOR, the cap domain undergoes a conformational change that will allow for specific cysteine residues to be able to open the epoxide ring and to recreate Vitamin K Quinone. The catalytic cycle begins in an open fully oxidized conformation. <scene name='90/904321/Openvkor/3'>Open Oxidized Conformation</scene> This conformation has slightly different parts. These include the Anchor (green), the cap region (blue), 3-4 Loop (pink), and luminal helix (yellow). When Vitamin K Epoxide binds, the entire cap domain undergoes a slight conformation change, but the luminal helix has a larger change. The luminal helix (yellow) bends forward where specific cysteines on this region are in proximity to other important cysteines. The luminal helix is then referred to as the beta hairpin (purple). <scene name='90/904321/Vkoclosed/3'>Partially Oxidized Closed Conformation</scene> |
==Significant Cysteines== | ==Significant Cysteines== | ||
Revision as of 19:29, 29 March 2022
Vitamin K Epoxide Reductase
| |||||||||||
References
- ↑ Ransey E, Paredes E, Dey SK, Das SR, Heroux A, Macbeth MR. Crystal structure of the Entamoeba histolytica RNA lariat debranching enzyme EhDbr1 reveals a catalytic Zn(2+) /Mn(2+) heterobinucleation. FEBS Lett. 2017 Jul;591(13):2003-2010. doi: 10.1002/1873-3468.12677. Epub 2017, Jun 14. PMID:28504306 doi:http://dx.doi.org/10.1002/1873-3468.12677
- ↑ Liu S, Li S, Shen G, Sukumar N, Krezel AM, Li W. Structural basis of antagonizing the vitamin K catalytic cycle for anticoagulation. Science. 2020 Nov 5. pii: science.abc5667. doi: 10.1126/science.abc5667. PMID:33154105 doi:http://dx.doi.org/10.1126/science.abc5667
- ↑ Olson RE. The function and metabolism of vitamin K. Annu Rev Nutr. 1984;4:281-337. doi: 10.1146/annurev.nu.04.070184.001433. PMID:6380538 doi:http://dx.doi.org/10.1146/annurev.nu.04.070184.001433
- ↑ Wu S, Chen X, Jin DY, Stafford DW, Pedersen LG, Tie JK. Warfarin and vitamin K epoxide reductase: a molecular accounting for observed inhibition. Blood. 2018 Aug 9;132(6):647-657. doi: 10.1182/blood-2018-01-830901. Epub 2018, May 9. PMID:29743176 doi:http://dx.doi.org/10.1182/blood-2018-01-830901
- ↑ Chatron N, Abi Khalil R, Benoit E, Lattard V. Structural Investigation of the Vitamin K Epoxide Reductase (VKORC1) Binding Site with Vitamin K. Biochemistry. 2020 Apr 7;59(13):1351-1360. doi: 10.1021/acs.biochem.9b01084. Epub, 2020 Mar 23. PMID:32182040 doi:http://dx.doi.org/10.1021/acs.biochem.9b01084
- ↑ Shen G, Cui W, Cao Q, Gao M, Liu H, Su G, Gross ML, Li W. The catalytic mechanism of vitamin K epoxide reduction in a cellular environment. J Biol Chem. 2021 Jan-Jun;296:100145. doi: 10.1074/jbc.RA120.015401. Epub 2020, Dec 10. PMID:33273012 doi:http://dx.doi.org/10.1074/jbc.RA120.015401