Sandbox Reserved 1725
From Proteopedia
(Difference between revisions)
| Line 17: | Line 17: | ||
===Catalytic Cysteines=== | ===Catalytic Cysteines=== | ||
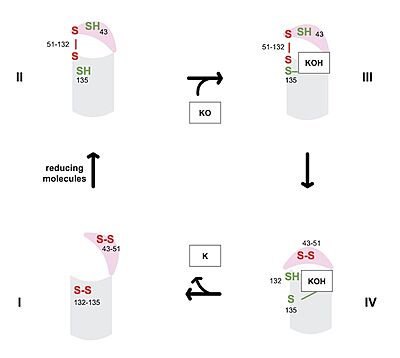
| - | [[Image:Vkor_cat_cycle_jpeg.jpg|400 px|right|thumb|Figure 3. Catalytic Cycle of VKOR]] | + | [[Image:Vkor_cat_cycle_jpeg.jpg|400 px|right|thumb|'''Figure 3. Catalytic Cycle of VKOR''' VKOR's luminal domain is represented by a the pink semicircle and the transmembrane domain is represented by the gray cylinder. State I displays the fully oxidized VKOR in an open conformation. State II is a partially oxidized VKOR with free cysteines 43 and 135. State III is cysteine 135 interacting with KOH or KH to induce a closed conformation. State IV shows cysteine 132 attacking cysteine 135-KOH or KH to generate K.]] |
| - | The catalytic cycle of VKOR includes transitions from open to closed conformations by means of disulfide bridge-induced conformational changes. The substituent cysteines ( | + | The catalytic cycle of VKOR includes transitions from open to closed conformations by means of disulfide bridge-induced conformational changes (Figure 3). Open conformations of VKOR exist when there is no ligand within the binding pocket. Closed conformations exist when some substrate exists within the binding pocket of VKOR. The substituent cysteines (Fig 3, step I) act as reducing agents for the substrate, which can be either Vitamin K epoxide (KO) or partially reduced Vitamin K. The first step of the catalytic cycle (Figure 3) is the wild type open conformation, <scene name='90/904329/Cat_cycle_i/1'>step I</scene>. This step is characterized by an open cap domain with disulfide bonds between cysteines 43 and 51 and between cysteines 132 and 135 <ref name="Liu">PMID:33154105</ref>. The second step of the catalytic cycle is a partially oxidized open conformation, <scene name='90/904329/Cat_cycle_2/1'>step II</scene>. This step is characterized by a disulfide bond between the luminal and transmembrane domain (Fig 3, step II). The transmembrane domain contains a free Cys135 and the luminal domain contains a free Cys43 <ref name="Liu">PMID:33154105</ref>. Step II is labeled as open because no ligand exists within its binding pocket despite the disulfide bridge that connects the luminal and transmembrane domains. The next step of the cycle, <scene name='90/904329/Cat_cycle_3/5'>step III</scene>, is also a closed structure with an intact disulfide bond between Cys51 and Cys132. Cys135 is not involved in a disulfide bridge and assists with substrate binding by forming a stable adduct with KOH or K. This binding induces the closed conformation and uses Cys43 in the luminal membrane for electron transfer <ref name="Liu">PMID:33154105</ref>. <scene name='90/904329/Cat_cycle_4/1'>Step IV</scene> of the catalytic cycle is the last closed conformation. The Cys51-Cys132 bond is broken as Cys43 bonds with Cys51, recreating the disulfide bridge pattern of the open state. Cys132 is then free to bond with Cys135, releasing the product that was bound to the Cys135. Overall the catalytic cycle of VKOR is dependent on open and closed conformational changes of the protein and ultimately is used to generate vitamin K from vitamin K epoxide <ref name="Liu">PMID:33154105</ref>. |
| + | ==== Warfarin Dependence on Catalytic Cysteines === | ||
The anticoagulant [https://en.wikipedia.org/wiki/Warfarin warfarin] works by inhibiting VKOR (See "Medical Relevance"). Warfarin binding also depends on the catalytic cysteines. Warfarin is able to bind to the fully oxidized open form of VKOR as shown in <scene name='90/904329/Cat_cycle_i/1'>step I</scene>. Once Warfarin binds, VKOR is considered to be in a closed conformation since the substrate cannot enter, despite the lack of disulfide bridge changes. Warfarin can also bind to the partially oxidized form of VKOR as shown in <scene name='90/904329/Cat_cycle_2/1'>step II</scene>. | The anticoagulant [https://en.wikipedia.org/wiki/Warfarin warfarin] works by inhibiting VKOR (See "Medical Relevance"). Warfarin binding also depends on the catalytic cysteines. Warfarin is able to bind to the fully oxidized open form of VKOR as shown in <scene name='90/904329/Cat_cycle_i/1'>step I</scene>. Once Warfarin binds, VKOR is considered to be in a closed conformation since the substrate cannot enter, despite the lack of disulfide bridge changes. Warfarin can also bind to the partially oxidized form of VKOR as shown in <scene name='90/904329/Cat_cycle_2/1'>step II</scene>. | ||
| - | === | + | === Key Substrate Binding Residues === |
| - | VKOR uses two catalytic amino acids, tyrosine 139 and asparagine 80, to stabilize <scene name='90/904329/Kohhbond/2'>vitamin K</scene> | + | VKOR uses two catalytic amino acids, tyrosine 139 and asparagine 80, to stabilize binding to all forms of <scene name='90/904329/Kohhbond/2'>vitamin K</scene> and <scene name='90/904329/Warfarinhbond/3'>vitamin K antagonists</scene>, such as Warfarin, in the binding pocket. Tyr139 and Asn80 hydrogen bond to carbonyl groups on both structures and stabilizes them within the binding pocket <ref name="Liu">PMID:33154105</ref>. |
=== Hydrophobic Interactions === | === Hydrophobic Interactions === | ||
| - | Other than the two previously mentioned hydrogen bonds (Tyr139 and Asn80), <scene name='90/904329/Kohhydrophobic/2'>vitamin K</scene> and <scene name='90/904329/Warfarinhydrophobic/1'>antagonists</scene> are bound | + | Other than the two previously mentioned hydrogen bonds (Tyr139 and Asn80), <scene name='90/904329/Kohhydrophobic/2'>vitamin K</scene> and <scene name='90/904329/Warfarinhydrophobic/1'> VKOR antagonists</scene> are bound via hydrophobic interactions within the binding pocket of VKOR. Hydrophobic residues of VKOR such as Phe80, Phe87, and Tyr88, form a hydrophobic tunnel within the binding pocket <ref name="Liu">PMID:33154105</ref>. |
== Medical Relevance == | == Medical Relevance == | ||
===Warfarin=== | ===Warfarin=== | ||
| - | Warfarin is the most widely prescribed oral anticoagulant | + | Warfarin is the most widely prescribed oral anticoagulant and targets blood clotting via inhibition of vitamin K epoxide reductase. The FDA approved uses for cardiac conditions (myocardial infarction, atrial fibrillation) as well as for deep vein thrombosis and pulmonary embolism. Due to the inhibition of the normal blood clotting cycle, patients taking warfarin are at risk for hemorrhage which can occur anywhere in the body. <ref name="Patel">PMID:29261922</ref> |
Warfarin is a <scene name='90/904330/Bothsubstratevkor/2'>structural mimic</scene> of Vitamin K and acts as a competitive inhibitor. There are around 30 known missense mutations that lead to warfarin resistance in patients, but these mutations do not affect Vitamin K binding for reasons which are not yet fully understood. Such patients require higher doses of warfarin to reach therapeutic level or require a different anticoagulant drug. <ref name="Wu">PMID:29743176</ref> | Warfarin is a <scene name='90/904330/Bothsubstratevkor/2'>structural mimic</scene> of Vitamin K and acts as a competitive inhibitor. There are around 30 known missense mutations that lead to warfarin resistance in patients, but these mutations do not affect Vitamin K binding for reasons which are not yet fully understood. Such patients require higher doses of warfarin to reach therapeutic level or require a different anticoagulant drug. <ref name="Wu">PMID:29743176</ref> | ||
Revision as of 19:54, 7 April 2022
| This Sandbox is Reserved from February 28 through September 1, 2022 for use in the course CH462 Biochemistry II taught by R. Jeremy Johnson at the Butler University, Indianapolis, USA. This reservation includes Sandbox Reserved 1700 through Sandbox Reserved 1729. |
To get started:
More help: Help:Editing |
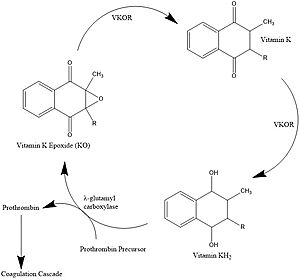
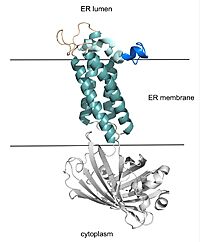
Vitamin K Epoxide Reductase
| |||||||||||
References
- ↑ 1.0 1.1 Stafford DW. The vitamin K cycle. J Thromb Haemost. 2005 Aug;3(8):1873-8. doi: 10.1111/j.1538-7836.2005.01419.x. PMID:16102054 doi:http://dx.doi.org/10.1111/j.1538-7836.2005.01419.x
- ↑ 2.0 2.1 Blanchard RA, Furie BC, Jorgensen M, Kruger SF, Furie B. Acquired vitamin K-dependent carboxylation deficiency in liver disease. N Engl J Med. 1981 Jul 30;305(5):242-8. doi: 10.1056/NEJM198107303050502. PMID:6165889 doi:http://dx.doi.org/10.1056/NEJM198107303050502
- ↑ Swanson JC, Suttie JW. Vitamin K dependent in vitro production of prothrombin. Biochemistry. 1982 Nov 9;21(23):6011-8. doi: 10.1021/bi00266a044. PMID:6758841 doi:http://dx.doi.org/10.1021/bi00266a044
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 Liu S, Li S, Shen G, Sukumar N, Krezel AM, Li W. Structural basis of antagonizing the vitamin K catalytic cycle for anticoagulation. Science. 2020 Nov 5. pii: science.abc5667. doi: 10.1126/science.abc5667. PMID:33154105 doi:http://dx.doi.org/10.1126/science.abc5667
- ↑ 5.0 5.1 Patel S, Singh R, Preuss CV, Patel N. Warfarin PMID:29261922
- ↑ Wu S, Chen X, Jin DY, Stafford DW, Pedersen LG, Tie JK. Warfarin and vitamin K epoxide reductase: a molecular accounting for observed inhibition. Blood. 2018 Aug 9;132(6):647-657. doi: 10.1182/blood-2018-01-830901. Epub 2018, May 9. PMID:29743176 doi:http://dx.doi.org/10.1182/blood-2018-01-830901
- ↑ Chong YK, Mak TW. Superwarfarin (Long-Acting Anticoagulant Rodenticides) Poisoning: from Pathophysiology to Laboratory-Guided Clinical Management. Clin Biochem Rev. 2019 Nov;40(4):175-185. doi: 10.33176/AACB-19-00029. PMID:31857739 doi:http://dx.doi.org/10.33176/AACB-19-00029
Student Contributors
Izabella Jordan, Emma Varness