We apologize for Proteopedia being slow to respond. For the past two years, a new implementation of Proteopedia has been being built. Soon, it will replace this 18-year old system. All existing content will be moved to the new system at a date that will be announced here.
Sandbox Reserved 1703
From Proteopedia
(Difference between revisions)
| Line 11: | Line 11: | ||
===Overall Structure=== | ===Overall Structure=== | ||
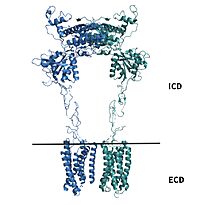
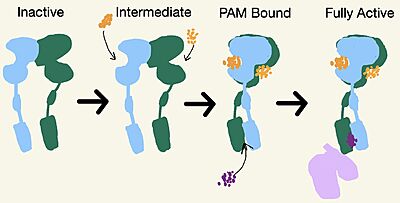
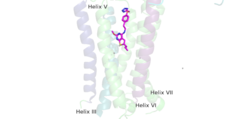
| - | [https://en.wikipedia.org/wiki/Cryogenic_electron_microscopy Cryo-EM] studies of mGlu2 have yielded adequate structural maps of mGlu2 in various activation states. These maps provided clearer understanding of the conformational changes between the inactive and active states of mGlu2<ref name="Lin" />. The overall <scene name='90/904307/Inactive_structure/1'>structure</scene> of the mGlu2 is composed of 3 main parts: a ligand binding <scene name='90/904307/Better_inactive_structure/3'>Venus Fly Trap Domain (VFT)</scene>, followed by a <scene name='90/904307/Better_inactive_structure/2'>Cysteine Rich Domain (CRD)</scene> linker to the <scene name='90/904307/Better_inactive_structure/4'>Transmembrane Domain</scene> that contains 7 | + | [https://en.wikipedia.org/wiki/Cryogenic_electron_microscopy Cryo-EM] studies of mGlu2 have yielded adequate structural maps of mGlu2 in various activation states. These maps provided clearer understanding of the conformational changes between the inactive and active states of mGlu2<ref name="Lin" />. The overall <scene name='90/904307/Inactive_structure/1'>structure</scene> of the mGlu2 is composed of 3 main parts: a ligand binding <scene name='90/904307/Better_inactive_structure/3'>Venus Fly Trap Domain (VFT)</scene>, followed by a <scene name='90/904307/Better_inactive_structure/2'>Cysteine Rich Domain (CRD)</scene> linker to the <scene name='90/904307/Better_inactive_structure/4'>Transmembrane Domain</scene> that contains 7 α-helices (7TM) on both the α and β chains. The VFT and CRD are located in the intracellular domain (ICD), while the TMD is located in the extracellular domain (ECD) (Figure 2). The TMD aids in the binding of the G-protein. |
[[Image:Domains of mGlu2.jpg|200 px|right|thumb|'''Figure 2.'''Show the regions of mGlu2.]] | [[Image:Domains of mGlu2.jpg|200 px|right|thumb|'''Figure 2.'''Show the regions of mGlu2.]] | ||
| - | mGlu2 is a [https://en.wikipedia.org/wiki/Protein_dimer homodimer]. Dimerization of mGlu2 is required to relay glutamate binding from the ECD to its TMD. The homodimer of mGlu2 contains an | + | mGlu2 is a [https://en.wikipedia.org/wiki/Protein_dimer homodimer]. Dimerization of mGlu2 is required to relay glutamate binding from the ECD to its TMD. The homodimer of mGlu2 contains an <scene name='90/904308/Alphaandbetachain/1'>α-chain and a β-chain</scene>. Occupation of both ECDs with the agonist, glutamate, is necessary for a fully active mGlu2<ref name="Du">Du, Juan, et al. “Structures of Human mglu2 and mglu7 Homo- and Heterodimers.” Nature News, Nature Publishing Group, 16 June 2021, https://www.nature.com/articles/s41586-021-03641-w.></ref>. However, only one chain in the dimer is responsible for activation of the G-protein, this suggests an asymmetrical signal transduction mechanism for mGlu2<ref name="Lin"/>. |
===Inactive State=== | ===Inactive State=== | ||
Revision as of 21:07, 18 April 2022
Metabotropic Glutamate Receptor 2
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Lin S, Han S, Cai X, Tan Q, Zhou K, Wang D, Wang X, Du J, Yi C, Chu X, Dai A, Zhou Y, Chen Y, Zhou Y, Liu H, Liu J, Yang D, Wang MW, Zhao Q, Wu B. Structures of Gi-bound metabotropic glutamate receptors mGlu2 and mGlu4. Nature. 2021 Jun;594(7864):583-588. doi: 10.1038/s41586-021-03495-2. Epub 2021, Jun 16. PMID:34135510 doi:http://dx.doi.org/10.1038/s41586-021-03495-2
- ↑ 2.0 2.1 Seven, Alpay B., et al. “G-Protein Activation by a Metabotropic Glutamate Receptor.” Nature News, Nature Publishing Group, 30 June 2021, https://www.nature.com/articles/s1586-021-03680-3
- ↑ Du, Juan, et al. “Structures of Human mglu2 and mglu7 Homo- and Heterodimers.” Nature News, Nature Publishing Group, 16 June 2021, https://www.nature.com/articles/s41586-021-03641-w.>
- ↑ 4.0 4.1 “Metabotropic Glutamate Receptor.” Wikipedia, Wikimedia Foundation, 27 Mar. 2022, https://en.wikipedia.org/wiki/Metabotropic_glutamate_receptor
- ↑ \“Schizophrenia.” National Institute of Mental Health, U.S. Department of Health and Human Services, https://www.nimh.nih.gov/health/topics/schizophrenia
- ↑ 6.0 6.1 Ellaithy A, Younkin J, Gonzalez-Maeso J, Logothetis DE. Positive allosteric modulators of metabotropic glutamate 2 receptors in schizophrenia treatment. Trends Neurosci. 2015 Aug;38(8):506-16. doi: 10.1016/j.tins.2015.06.002. Epub, 2015 Jul 4. PMID:26148747 doi:http://dx.doi.org/10.1016/j.tins.2015.06.002
- ↑ 7.0 7.1 7.2 7.3 Muguruza C, Meana JJ, Callado LF. Group II Metabotropic Glutamate Receptors as Targets for Novel Antipsychotic Drugs. Front Pharmacol. 2016 May 20;7:130. doi: 10.3389/fphar.2016.00130. eCollection, 2016. PMID:27242534 doi:http://dx.doi.org/10.3389/fphar.2016.00130
Student Contributors
Frannie Brewer and Ashley Wilkinson