This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1715
From Proteopedia
(Difference between revisions)
| Line 44: | Line 44: | ||
== Conformational Changes == | == Conformational Changes == | ||
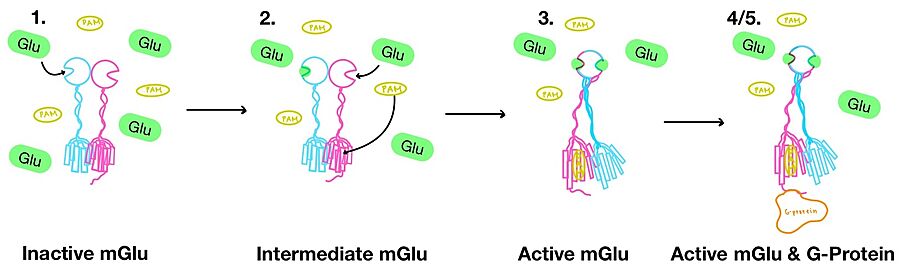
| - | [[Image:Overview_mGlu_2.jpg|900 px|center|thumb|Figure 3. Illustration of mGlu's conformational change process.]] | ||
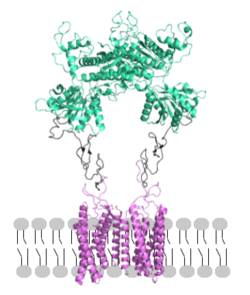
'''1.''' In its resting state, mGlu is in an <scene name='90/904320/Inactive_mglu2_first_picture/6'>inactive homodimeric form</scene>. In this conformation, the receptor is considered open with an inter-lobe angle of 44°<ref name="Seven">PMID:34194039</ref>. The structure has two free glutamate binding sites in the VFT, the CRDs are separated, and the TMD is not interacting with a G protein<ref name="Seven">PMID:34194039</ref>. | '''1.''' In its resting state, mGlu is in an <scene name='90/904320/Inactive_mglu2_first_picture/6'>inactive homodimeric form</scene>. In this conformation, the receptor is considered open with an inter-lobe angle of 44°<ref name="Seven">PMID:34194039</ref>. The structure has two free glutamate binding sites in the VFT, the CRDs are separated, and the TMD is not interacting with a G protein<ref name="Seven">PMID:34194039</ref>. | ||
'''2.''' In the intermediate activation state, also known as the open-closed conformation, one glutamate is bound in one binding pocket of VFT. This single <scene name='90/904320/Mglu_binding/9'>glutamate bound state</scene> is still considered inactive as the receptor has not changed the conformations in the CRD and thus the TMD. With the same asymmetric transmembrane helices formation, a <scene name='90/904320/Inactive_tmd_interface/1'>TM3-TM4 interface</scene> is still present and mGlu cannot interact with a G protein<ref name="Seven">PMID:34194039</ref>. | '''2.''' In the intermediate activation state, also known as the open-closed conformation, one glutamate is bound in one binding pocket of VFT. This single <scene name='90/904320/Mglu_binding/9'>glutamate bound state</scene> is still considered inactive as the receptor has not changed the conformations in the CRD and thus the TMD. With the same asymmetric transmembrane helices formation, a <scene name='90/904320/Inactive_tmd_interface/1'>TM3-TM4 interface</scene> is still present and mGlu cannot interact with a G protein<ref name="Seven">PMID:34194039</ref>. | ||
| - | [[Image: | + | [[Image:Overview_mGlu_2.jpg|900 px|center|thumb|Figure 3. Illustration of mGlu's conformational change process.]] |
| + | |||
'''3.''' A second glutamate then binds to the other <scene name='90/904320/Active_site_interactions/4'>binding pocket</scene> of the VFT. Mediated by L639, F643, N735, W773, and F776, a <scene name='90/904320/Pam/8'>positive allosteric modulator</scene> (PAM) also binds within the seven TMD helices of the alpha chain <ref name="Seven">PMID:34194039</ref>. This closed conformation of the VFT now has an inter-lobe angle of 25° is considered to be in the <scene name='90/904320/Active_mglu/10'>active conformation</scene><ref name="Seven">PMID:34194039</ref>. The binding of these ligands allows the CRDs to compact and come together. This transformation causes the TMD to form a separate, active asymmetric conformation with a <scene name='90/904320/Active_helices/14'>TM6-TM6 interface</scene> between the chains<ref name="Seven">PMID:34194039</ref>. | '''3.''' A second glutamate then binds to the other <scene name='90/904320/Active_site_interactions/4'>binding pocket</scene> of the VFT. Mediated by L639, F643, N735, W773, and F776, a <scene name='90/904320/Pam/8'>positive allosteric modulator</scene> (PAM) also binds within the seven TMD helices of the alpha chain <ref name="Seven">PMID:34194039</ref>. This closed conformation of the VFT now has an inter-lobe angle of 25° is considered to be in the <scene name='90/904320/Active_mglu/10'>active conformation</scene><ref name="Seven">PMID:34194039</ref>. The binding of these ligands allows the CRDs to compact and come together. This transformation causes the TMD to form a separate, active asymmetric conformation with a <scene name='90/904320/Active_helices/14'>TM6-TM6 interface</scene> between the chains<ref name="Seven">PMID:34194039</ref>. | ||
| + | |||
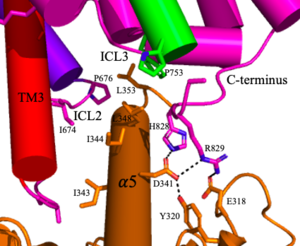
| + | [[Image:Screen Shot 2022-04-18 at 10.20.26 PM.png|300 px|right|thumb|Figure 4. The interaction between an active mGlu (magenta/lime/purple/crimson) and a G-protein (orange). Hydrogen bonds are shown through black dashes]] | ||
'''4.''' The crossover of the helices from the alpha and beta chains allows for intracellular loop 2 (ICL2) and the C-terminus to be properly ordered to interact with a single G protein<ref name="Seven">PMID:34194039</ref>. While hydrogen bonding is present between the C-terminus and alpha helix 5 of the G-protein, this <scene name='90/904320/Active_mglu/9'>mGlu/G-protein coupling</scene> is primarily driven by the hydrophobic interactions in the interface with the ɑ5 helix of the G protein<ref name="Seven">PMID:34194039</ref>(Figure 4). This coupling can only occur in the presence of a <scene name='90/904320/Pam/8'>PAM</scene> as the pocket in which the coupling occurs would be completely closed in its absence<ref name="Seven">PMID:34194039</ref>. | '''4.''' The crossover of the helices from the alpha and beta chains allows for intracellular loop 2 (ICL2) and the C-terminus to be properly ordered to interact with a single G protein<ref name="Seven">PMID:34194039</ref>. While hydrogen bonding is present between the C-terminus and alpha helix 5 of the G-protein, this <scene name='90/904320/Active_mglu/9'>mGlu/G-protein coupling</scene> is primarily driven by the hydrophobic interactions in the interface with the ɑ5 helix of the G protein<ref name="Seven">PMID:34194039</ref>(Figure 4). This coupling can only occur in the presence of a <scene name='90/904320/Pam/8'>PAM</scene> as the pocket in which the coupling occurs would be completely closed in its absence<ref name="Seven">PMID:34194039</ref>. | ||
Revision as of 06:52, 19 April 2022
Metabotropic Glutamate Receptor
| |||||||||||
Student Contributors
- Courtney Vennekotter
- Cade Chezem