This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Ceramidase
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
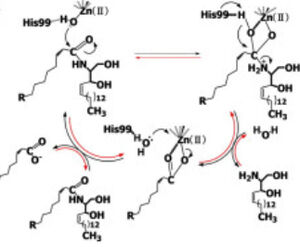
[[Image:CerN Mechanism Okino2009.jpg|300px|right|thumb|'''Figure 1''' Proposed mechanisms of the zinc-dependent hydrolysis of C2-ceramide (Black arrows) and the zinc-dependent synthesis of C2-ceramide from palmitate and sphingosine (Red arrows) by CerN . Figure adapted from Inoe et al.(2009)<ref name="Inoue">PMID:19088069</ref>]] | [[Image:CerN Mechanism Okino2009.jpg|300px|right|thumb|'''Figure 1''' Proposed mechanisms of the zinc-dependent hydrolysis of C2-ceramide (Black arrows) and the zinc-dependent synthesis of C2-ceramide from palmitate and sphingosine (Red arrows) by CerN . Figure adapted from Inoe et al.(2009)<ref name="Inoue">PMID:19088069</ref>]] | ||
'''CerN''' is an enzyme that catalyzes the cleavage of the [https://en.wikipedia.org/wiki/Sphingolipid Sphingolipid] <scene name='91/910024/Ceramide/3'>Ceramide</scene> at the <scene name='91/910024/Ceramide18/1'>N-acyl linkage</scene>, producing <scene name='91/910024/Ceramide/4'>sphingosine and a fatty acid</scene>.<ref name="Okino">PMID:9603946</ref> <ref name="Inoue">PMID:19088069</ref> CerN cleaves the N-acyl linkage within ceramides via '''zinc-dependent hydrolysis''' and the enzyme is also capable of '''synthesizing ceramide''' from sphingosine and palmitic acid by the reverse mechanism.<ref name="Inoue">PMID:19088069</ref><ref name="Reverse">PMID:10832092</ref> The zinc ion within the <scene name='91/910024/Activesite2/3'>active site</scene> is coordinated by His97, His204, Glu411, Tyr448, and a water molecule. His97 and Tyr448 are required for zinc binding within the active site. ''Ligand binding within the active site is recognized by Gly25, His99, Arg160, and Tyr460''.<ref name="Inoue">PMID:19088069</ref> Ser27 and Gly25 stabilize ceramide within the active site by forming a water-mediated hydrogen bond with the central OH of ceramide, and the carbonyl oxygen is stabilized by the Tyr448 and Tyr460. | '''CerN''' is an enzyme that catalyzes the cleavage of the [https://en.wikipedia.org/wiki/Sphingolipid Sphingolipid] <scene name='91/910024/Ceramide/3'>Ceramide</scene> at the <scene name='91/910024/Ceramide18/1'>N-acyl linkage</scene>, producing <scene name='91/910024/Ceramide/4'>sphingosine and a fatty acid</scene>.<ref name="Okino">PMID:9603946</ref> <ref name="Inoue">PMID:19088069</ref> CerN cleaves the N-acyl linkage within ceramides via '''zinc-dependent hydrolysis''' and the enzyme is also capable of '''synthesizing ceramide''' from sphingosine and palmitic acid by the reverse mechanism.<ref name="Inoue">PMID:19088069</ref><ref name="Reverse">PMID:10832092</ref> The zinc ion within the <scene name='91/910024/Activesite2/3'>active site</scene> is coordinated by His97, His204, Glu411, Tyr448, and a water molecule. His97 and Tyr448 are required for zinc binding within the active site. ''Ligand binding within the active site is recognized by Gly25, His99, Arg160, and Tyr460''.<ref name="Inoue">PMID:19088069</ref> Ser27 and Gly25 stabilize ceramide within the active site by forming a water-mediated hydrogen bond with the central OH of ceramide, and the carbonyl oxygen is stabilized by the Tyr448 and Tyr460. | ||
| - | Upon ligand binding, CerN enters the <scene name='91/910024/Closedsurf_use/1'>closed</scene> conformation. <ref name="Inoue">PMID:19088069</ref> ''His99 and Arg160 function in the catalysis of ceramide hydrolysis'', as they deprotonate their coordinated water molecule to produce a hydroxide ion. The carbonyl carbon of ceramide undergoes a '''nucleophilic attack''' by the hydroxide ion ('''Figure 1'''). The carbonyl oxygen stabilized by Tyr448 and Tyr460 is then passed to the zinc ion, allowing for the breakage of the N-acyl linkage.<ref name="Inoue">PMID:19088069</ref> Sphingosine is then released from the active site while the fatty acid remains bound to the zinc ion until it is replaced by a new water molecule, shifting CerN into the <scene name='91/910024/Activesite_open_surf/2'>open</scene> conformation. The synthesis of ceramide from palmitate and sphingosine occurs via the same mechanism but in reverse. <ref name="Inoue">PMID:19088069</ref> | + | Upon ligand binding, CerN enters the <scene name='91/910024/Closedsurf_use/1'>closed</scene> conformation. <ref name="Inoue">PMID:19088069</ref> ''His99 and Arg160 function in the catalysis of ceramide hydrolysis'', as they deprotonate their coordinated water molecule to produce a hydroxide ion. The carbonyl carbon of ceramide undergoes a '''nucleophilic attack''' by the hydroxide ion ('''Figure 1'''). The carbonyl oxygen stabilized by Tyr448 and Tyr460 is then passed to the zinc ion, allowing for the breakage of the N-acyl linkage.<ref name="Inoue">PMID:19088069</ref> Sphingosine is then released from the active site while the fatty acid remains bound to the zinc ion until it is replaced by a new water molecule, shifting CerN into the <scene name='91/910024/Activesite_open_surf/2'>open</scene> conformation. The synthesis of ceramide from palmitate and sphingosine occurs via the same mechanism but in reverse ('''Figure 1'''). <ref name="Inoue">PMID:19088069</ref> |
Revision as of 17:11, 30 April 2022
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 Inoue T, Okino N, Kakuta Y, Hijikata A, Okano H, Goda HM, Tani M, Sueyoshi N, Kambayashi K, Matsumura H, Kai Y, Ito M. Mechanistic insights into the hydrolysis and synthesis of ceramide by neutral ceramidase. J Biol Chem. 2009 Apr 3;284(14):9566-77. Epub 2008 Dec 16. PMID:19088069 doi:10.1074/jbc.M808232200
- ↑ 2.0 2.1 2.2 Okino N, Tani M, Imayama S, Ito M. Purification and characterization of a novel ceramidase from Pseudomonas aeruginosa. J Biol Chem. 1998 Jun 5;273(23):14368-73. PMID:9603946
- ↑ 3.0 3.1 Kita K, Okino N, Ito M. Reverse hydrolysis reaction of a recombinant alkaline ceramidase of Pseudomonas aeruginosa. Biochim Biophys Acta. 2000 May 31;1485(2-3):111-20. doi:, 10.1016/s1388-1981(00)00029-9. PMID:10832092 doi:http://dx.doi.org/10.1016/s1388-1981(00)00029-9