This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox reserved 1752
From Proteopedia
| Line 63: | Line 63: | ||
<Structure load='4g7O' size='350' frame='true' align='right' caption='Insert caption here' scene='Insert optional scene name here' /> | <Structure load='4g7O' size='350' frame='true' align='right' caption='Insert caption here' scene='Insert optional scene name here' /> | ||
| - | UvrD, also known as Helicase II, is one of many components responsible in repairing DNA damage. UvrD is involved in nucleotide excision repair (NER) and transcription-coupled DNA repair (TCR repair). TCR repair occurs during transcription when Helicase II binds to a RNA polymerase (RNAP) and slides backwards in order to fix the mismatched nucleotide. One of the main structural components of how UvrD binds to RNAP is by having a Tudor-domain like fold. This helps better explain the UvrD-RNAP interaction, which is a better explanation than just having nucleic-acid affinity. A main cause in damage to DNA, which requires for nucleotide excision repair is from UV radiation, which can come from multiple sources, including sunlight and UV light from tanning beds. | + | <scene name='92/925553/Uvrd/1'>UvrD</scene>, also known as Helicase II, is one of many components responsible in repairing DNA damage. UvrD is involved in nucleotide excision repair (NER) and transcription-coupled DNA repair (TCR repair). TCR repair occurs during transcription when Helicase II binds to a RNA polymerase (RNAP) and slides backwards in order to fix the mismatched nucleotide. One of the main structural components of how UvrD binds to RNAP is by having a Tudor-domain like fold. This helps better explain the UvrD-RNAP interaction, which is a better explanation than just having nucleic-acid affinity. A main cause in damage to DNA, which requires for nucleotide excision repair is from UV radiation, which can come from multiple sources, including sunlight and UV light from tanning beds. |
== UvrD Binding Site for DNA == | == UvrD Binding Site for DNA == | ||
Revision as of 03:34, 4 October 2022
Contents |
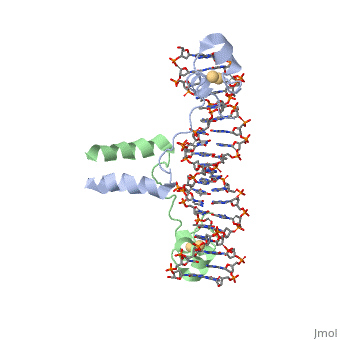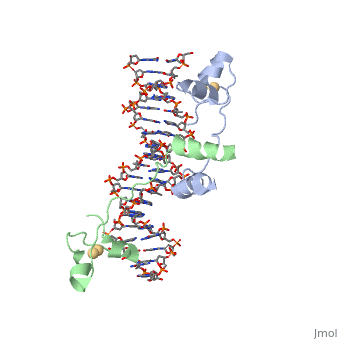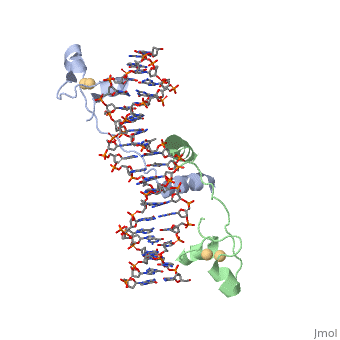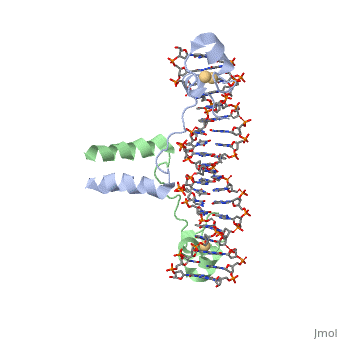
DNA RECOGNITION BY GAL4: STRUCTURE OF A PROTEIN/DNA COMPLEX
| |||||||||||
UvrD
|
, also known as Helicase II, is one of many components responsible in repairing DNA damage. UvrD is involved in nucleotide excision repair (NER) and transcription-coupled DNA repair (TCR repair). TCR repair occurs during transcription when Helicase II binds to a RNA polymerase (RNAP) and slides backwards in order to fix the mismatched nucleotide. One of the main structural components of how UvrD binds to RNAP is by having a Tudor-domain like fold. This helps better explain the UvrD-RNAP interaction, which is a better explanation than just having nucleic-acid affinity. A main cause in damage to DNA, which requires for nucleotide excision repair is from UV radiation, which can come from multiple sources, including sunlight and UV light from tanning beds.