This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Hemoglobin
From Proteopedia
| Line 3: | Line 3: | ||
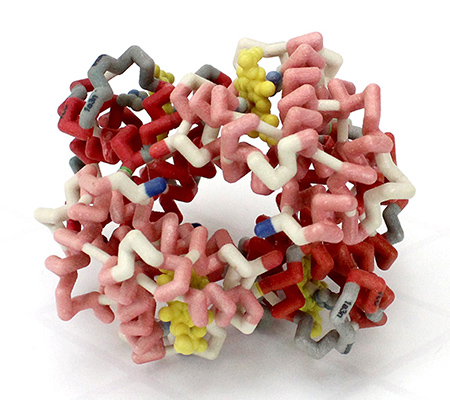
'''Hemoglobin''' is an oxygen-transport protein. Hemoglobin is an allosteric protein. It is a <scene name='32/32/Subunits_1hho/1'>tetramer</scene> composed of two types of subunits designated α and β, with stoichiometry <scene name='Hemoglobin/Alpha2beta2/7'>α2β2</scene>. The <scene name='Hemoglobin/Foursubunits/5'>four subunits</scene> of hemoglobin sit roughly at the corners of a tetrahedron, facing each other across a <scene name='Hemoglobin/Cavity/9'>cavity</scene> at the center of the molecule. Each of the subunits <scene name='Hemoglobin/Bbsubunitswithheme/5'>contains a heme</scene> prosthetic group. The <scene name='Hemoglobin/4heme/3'>heme molecules</scene> give hemoglobin its red color. | '''Hemoglobin''' is an oxygen-transport protein. Hemoglobin is an allosteric protein. It is a <scene name='32/32/Subunits_1hho/1'>tetramer</scene> composed of two types of subunits designated α and β, with stoichiometry <scene name='Hemoglobin/Alpha2beta2/7'>α2β2</scene>. The <scene name='Hemoglobin/Foursubunits/5'>four subunits</scene> of hemoglobin sit roughly at the corners of a tetrahedron, facing each other across a <scene name='Hemoglobin/Cavity/9'>cavity</scene> at the center of the molecule. Each of the subunits <scene name='Hemoglobin/Bbsubunitswithheme/5'>contains a heme</scene> prosthetic group. The <scene name='Hemoglobin/4heme/3'>heme molecules</scene> give hemoglobin its red color. | ||
| - | Each individual <scene name='Hemoglobin/Deoxyheme/8'>heme</scene> molecule contains one <scene name='Hemoglobin/Deoxyheme_fe/9'> | + | Each individual <scene name='Hemoglobin/Deoxyheme/8'>heme</scene> molecule contains one <scene name='Hemoglobin/Deoxyheme_fe/9'>Fe2+</scene> atom. In the lungs, where oxygen is abundant, an <scene name='Hemoglobin/Oxyheme_fe/7'>oxygen molecule</scene> binds to the ferrous iron atom of the heme molecule and is later released in tissues needing oxygen. The heme group binds oxygen while still attached to the <scene name='Hemoglobin/Oxysubunit/8'>hemoglobin monomer</scene>. The spacefill view of the hemoglobin polypeptide subunit with an oxygenated heme group shows how the <scene name='Hemoglobin/Oxysubunitsf/4'>oxygenated heme group is held</scene> within the polypeptide. |
<scene name='Hemoglobin/Anchortrace/5'>Anchoring of the heme</scene> is facilitated by a histidine nitrogen that binds to the iron. A second histidine is near the bound oxygen. The "arms" (propanoate groups) of the heme are hydrophilic and face the surface of the protein while the hydrophobic portions of the heme are buried among the hydrophobic amino acids of the protein. | <scene name='Hemoglobin/Anchortrace/5'>Anchoring of the heme</scene> is facilitated by a histidine nitrogen that binds to the iron. A second histidine is near the bound oxygen. The "arms" (propanoate groups) of the heme are hydrophilic and face the surface of the protein while the hydrophobic portions of the heme are buried among the hydrophobic amino acids of the protein. | ||
Current revision
| |||||||||||
References, for further information on Hemoglobin
To the structures used here:
- Baldwin (1980) "The crystal structure of human carbonmonoxy haemoglobin at 2.7A resolution", J. Mol. Biol. 136: 103. (1hco) PMID: 7373648
- Fermi, Perutz, Shaanan, & Fourme (1984) "The crystal structure of human deoxy haemoglobin at 1.74A resolution", J. Mol. Biol. 175: 159. (3hhb)
- Jotaro Igarashi, Kazuo Kobayashi and Ariki Matsuoka (2011) "A hydrogen-bonding network formed by the B10-E7-E11 residues of a truncated hemoglobin from Tetrahymena pyriformis is critical for stability of bound oxygen and nitric oxide detoxification", J. Biol. Inorg. Chem. 16(4):599-609 (3aq9) PMID: 21298303
General treatments of Hb allostery:
- Perutz (1970) "Stereochemistry of cooperative effects in haemoglobin", Nature 228: 726
- Baldwin & Chothia (1979) "Haemoglobin. The structural changes related to ligand binding and its allosteric mechanism", J. Mol. Biol. 129: 175. link
- Dickerson & Geis (1983) "Hemoglobin: Structure, Function, and Pathology", Benjamin/Cummings Publ., Menlo Park, CA
- Perutz (1989) "Mechanisms of cooperativity and allosteric regulation in proteins", Quarterly Rev. of Biophys. 22: 139-236
- Ackers, Doyle, Myers, & Daugherty (1992) "Molecular code for cooperativity in hemoglobin", Science 255: 54
- Perutz, Fermi, Poyart, Pagnier, & Kister (1993) "A novel allosteric mechanism in haemoglobin: Structure of bovine deoxyhaemoglobin, absence of specific chloride binding sites, and origin of the chloride-linked Bohr Effect in bovine and human haemoglobin", J. Mol. Biol. 233: 536
Hb structures in other quaternary states or intermediates:
- Silva, Rogers, & Arnone (1992) "A third quaternary structure of human hemoglobin A at 1.7A resolution", J. Biol. Chem. 267: 17248
- Smith, Lattman, & Carter (1991) "The mutation β99 Asp-Tyr stabilizes Y - A new, composite quaternary state of human hemoglobin", Proteins: Struct., Funct., Genet. 10: 81
- Liddington, Derewenda, Dodson, Hubbard, & Dodson (1992) "High resolution crystal structures and comparisons of T state deoxyhaemoglobin and two liganded T-state haemoglobins: T(α-oxy)haemoglobin and T(met)Haemoglobin", J. Mol. Biol. 228: 551
More information on hemoglobin
- Perutz, M.F. (1978) Hemoglobin Structure and Respiratory Transport, Scientific American, volume 239, number 6.
- Squires, J.E. (2002) Artificial Blood, Science 295, 1002.
- Vichinsky, E. (2002) New therapies in sickle cell disease. Lancet 24, 629.
Content Donators
Currently (June 22, 2008) most all of the content of this page comes from three main sources of generously donated content. Their work has been imported into this page. In their order of appearance on the page:
- Content adapted with permission from Eric Martz's hemoglobin tutorial at http://molviz.org
- Content adapted with permission from David S. Goodsell and Shuchismita Dutta's Molecule of the Month on Hemoglobin http://mgl.scripps.edu/people/goodsell/pdb/pdb41/pdb41_1.html
- Content adapted with permission from Jane S. and David C. Richardson's http://kinemage.biochem.duke.edu/
Proteopedia Page Contributors and Editors (what is this?)
Eran Hodis, Michal Harel, Joel L. Sussman, Alexander Berchansky, Jaime Prilusky, Eric Martz, Karl Oberholser, Tihitina Y Aytenfisu, Mark Hoelzer, Marc Gillespie, Ann Taylor, Hannah Campbell, Karsten Theis