This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1779
From Proteopedia
(Difference between revisions)
| Line 19: | Line 19: | ||
The ECD of TSHR is anchored to the membrane through seven transmembrane helices (7TMD), characteristic of GPCRs. Conformational changes in the 7TMD activate intracellular G-protein signaling<ref name= "Keinau et al.">Kleinau, G., Worth, C. L., Kreuchwig, A., Biebermann, H., Marcinkowski, P., Scheerer, P., & Krause, G. (2017). Structural–functional features of the thyrotropin receptor: A class A G-protein-coupled receptor at work. Frontiers in Endocrinology, 8. https://doi.org/10.3389/fendo.2017.00086</ref>. Once TSH binds, changes to the p10 peptide are transmitted to the 7TMD. Specifically, hinge helix rotation causes displacement of the p10 peptide that allows the <scene name='95/952709/Helix_7_of_7tmd/2'>seventh transmembrane helix (TM7)</scene> to migrate towards the center of the 7TMD to increase van Der Waals contacts. Additionally, K660 of TM7 forms a stabilizing <scene name='95/952709/Helix_7_and__p10_interaction/6'>ionic interaction</scene> with E409 of the p10 region. Hinge helix movement also rearranges Y279 relative to I486 on the neighboring <scene name='95/952709/Hinge_helix_ecl1/3'>extracellular loop 1 (ECL1) helix</scene>, which links two transmembrane helices and is located extracellularly. Substitution of these residues leads to substantial shifts in the activation of the thyrotropin receptor. Structurally guided mutagenic studies have shown that replacing isoleucine with a more sizeable phenylalanine decreases TSH signaling potency<ref name="Faust et al.">Faust, B., Billesbølle, C.B., Suomivuori, CM. et al. Autoantibody mimicry of hormone action at the thyrotropin receptor. Nature 609, 846–853 (2022). https://doi.org/10.1038/s41586-022-</ref><ref name="Vlaeminck-Guillem et al.">Virginie Vlaeminck-Guillem, Su-Chin Ho, Patrice Rodien, Gilbert Vassart, Sabine Costagliola, Activation of the cAMP Pathway by the TSH Receptor Involves Switching of the Ectodomain from a Tethered Inverse Agonist to an Agonist, Molecular Endocrinology, Volume 16, Issue 4, 1 April 2002, Pages 736–746, https://doi.org/10.1210/mend.16.4.0816</ref>.The sixth transmembrane helix of TSHR moves outward from the center of the 7TMD to facilitate α-helix 5 of the α-subunit of the G protein (Gα)<ref name="Faust et al.">Faust, B., Billesbølle, C.B., Suomivuori, CM. et al. Autoantibody mimicry of hormone action at the thyrotropin receptor. Nature 609, 846–853 (2022). https://doi.org/10.1038/s41586-022-</ref><ref name="Goricanec et al.">Goricanec, D., Stehle, R., Egloff, P., Grigoriu, S., Plückthun, A., Wagner, G., & Hagn, F. (2016). Conformational dynamics of a G-protein α subunit is tightly regulated by nucleotide binding. Proceedings of the National Academy of Sciences, 113(26). https://doi.org/10.1073/pnas.1604125113 </ref>. Gα is activated for intracellular signaling when GDP is exchanged for GTP and dissociates from the γ- and β-subunits of the G-protein (Gγ and Gβ) to bind with other target proteins. Activation of the Gα is caused by conformational shifts in the 7TMD and three intracellular loops which directly interact with the G-protein<ref name= "Keinau et al.">Kleinau, G., Worth, C. L., Kreuchwig, A., Biebermann, H., Marcinkowski, P., Scheerer, P., & Krause, G. (2017). Structural–functional features of the thyrotropin receptor: A class A G-protein-coupled receptor at work. Frontiers in Endocrinology, 8. https://doi.org/10.3389/fendo.2017.00086</ref>. Conformational shifts in transmembrane helices are the mechanism of changing interactions of the G-protein with the receptor. Activation of the G-protein is caused by these conformational shifts. | The ECD of TSHR is anchored to the membrane through seven transmembrane helices (7TMD), characteristic of GPCRs. Conformational changes in the 7TMD activate intracellular G-protein signaling<ref name= "Keinau et al.">Kleinau, G., Worth, C. L., Kreuchwig, A., Biebermann, H., Marcinkowski, P., Scheerer, P., & Krause, G. (2017). Structural–functional features of the thyrotropin receptor: A class A G-protein-coupled receptor at work. Frontiers in Endocrinology, 8. https://doi.org/10.3389/fendo.2017.00086</ref>. Once TSH binds, changes to the p10 peptide are transmitted to the 7TMD. Specifically, hinge helix rotation causes displacement of the p10 peptide that allows the <scene name='95/952709/Helix_7_of_7tmd/2'>seventh transmembrane helix (TM7)</scene> to migrate towards the center of the 7TMD to increase van Der Waals contacts. Additionally, K660 of TM7 forms a stabilizing <scene name='95/952709/Helix_7_and__p10_interaction/6'>ionic interaction</scene> with E409 of the p10 region. Hinge helix movement also rearranges Y279 relative to I486 on the neighboring <scene name='95/952709/Hinge_helix_ecl1/3'>extracellular loop 1 (ECL1) helix</scene>, which links two transmembrane helices and is located extracellularly. Substitution of these residues leads to substantial shifts in the activation of the thyrotropin receptor. Structurally guided mutagenic studies have shown that replacing isoleucine with a more sizeable phenylalanine decreases TSH signaling potency<ref name="Faust et al.">Faust, B., Billesbølle, C.B., Suomivuori, CM. et al. Autoantibody mimicry of hormone action at the thyrotropin receptor. Nature 609, 846–853 (2022). https://doi.org/10.1038/s41586-022-</ref><ref name="Vlaeminck-Guillem et al.">Virginie Vlaeminck-Guillem, Su-Chin Ho, Patrice Rodien, Gilbert Vassart, Sabine Costagliola, Activation of the cAMP Pathway by the TSH Receptor Involves Switching of the Ectodomain from a Tethered Inverse Agonist to an Agonist, Molecular Endocrinology, Volume 16, Issue 4, 1 April 2002, Pages 736–746, https://doi.org/10.1210/mend.16.4.0816</ref>.The sixth transmembrane helix of TSHR moves outward from the center of the 7TMD to facilitate α-helix 5 of the α-subunit of the G protein (Gα)<ref name="Faust et al.">Faust, B., Billesbølle, C.B., Suomivuori, CM. et al. Autoantibody mimicry of hormone action at the thyrotropin receptor. Nature 609, 846–853 (2022). https://doi.org/10.1038/s41586-022-</ref><ref name="Goricanec et al.">Goricanec, D., Stehle, R., Egloff, P., Grigoriu, S., Plückthun, A., Wagner, G., & Hagn, F. (2016). Conformational dynamics of a G-protein α subunit is tightly regulated by nucleotide binding. Proceedings of the National Academy of Sciences, 113(26). https://doi.org/10.1073/pnas.1604125113 </ref>. Gα is activated for intracellular signaling when GDP is exchanged for GTP and dissociates from the γ- and β-subunits of the G-protein (Gγ and Gβ) to bind with other target proteins. Activation of the Gα is caused by conformational shifts in the 7TMD and three intracellular loops which directly interact with the G-protein<ref name= "Keinau et al.">Kleinau, G., Worth, C. L., Kreuchwig, A., Biebermann, H., Marcinkowski, P., Scheerer, P., & Krause, G. (2017). Structural–functional features of the thyrotropin receptor: A class A G-protein-coupled receptor at work. Frontiers in Endocrinology, 8. https://doi.org/10.3389/fendo.2017.00086</ref>. Conformational shifts in transmembrane helices are the mechanism of changing interactions of the G-protein with the receptor. Activation of the G-protein is caused by these conformational shifts. | ||
| - | === Leucine Rich Repeats === | ||
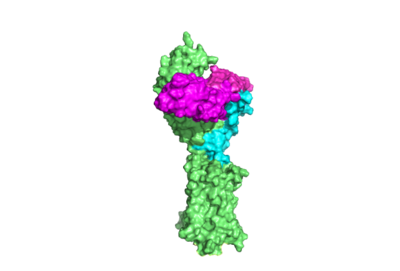
| - | The Leucine Rich Repeat Domain (LRRD) is part of the <scene name='95/952708/Tshr_chainr_ecd/1'>ECD</scene> of TSHR and contains <scene name='95/952707/Lrr/3'>10-11 Leucine Rich Repeats</scene>. A unique feature of this region is that it is composed entirely of β-pleated sheets. These β-pleated sheets of the LRRD provides a concave binding surface for TSH, including <scene name='95/952707/Interactions_with_thyrotropin/2'>K209 and K58</scene> <ref name="Duan et al.">PMID: 35940204</ref>. These interact with <scene name='95/952707/Interactions_with_thyrotropin/2'>N91 and E98</scene> in the seatbelt region of TSH forming a salt bridge and initiating the conformational change by pulling on the hinge region of the receptor <ref name="Faust">PMID: 35940205</ref>. This interaction is specific to TSH and TSHR. When other agonists or antagonists bind to the receptor, the change in conformation is a result of different residues interacting, as explained later in the page. The Leucine residues in the LRRD determine ECD folding and which residues are located on the exterior protein and interacting with ligands. | ||
| - | |||
| - | === Active and Inactive Form === | ||
| - | [[Image:Morph_pics2.png|200 px|right|thumb|Figure 2: Inactive form of the thyrotropin receptor shown in blue. Active form of the thyrotropin receptor shown in green.]] | ||
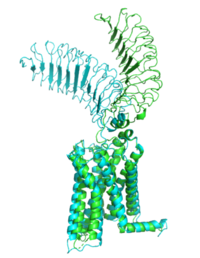
| - | The TSHR protein exists in two states: active and inactive (Figure 2) (GREEN LINK ?). The <scene name='95/952708/Tshr_chainr_ecd/1'>ECD</scene> protrudes from the cell membrane into the space outside the cell. The <scene name='95/952708/Tshr_chainr_tm/1'>transmembrane domain</scene> contains 7 alpha helices that reside within the cell membrane. The <scene name='95/952708/Tshr_chainr/4'>TSHR active form</scene> exists when bound to the <scene name='95/952708/Tsh_7t9i/1'>TSH</scene>. One proposed mechanism for the transition from the active to inactive describes that in a natural state, the TSHR ECD can spontaneously transition to the up state, leading to constitutive activity. In this active state, TSH will bind and keep the active state in the up position because of clash with the cell membrane.<ref name="Faust" /> Conformational change of ECD allows for signal transduction through the TM and into the cell. The ECD rotates 55 degrees up in the active form. <ref name="Faust" /> | ||
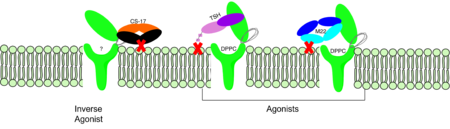
== TSHR Agonists and Antagonists == | == TSHR Agonists and Antagonists == | ||
Revision as of 20:49, 12 April 2023
>
| This Sandbox is Reserved from February 27 through August 31, 2023 for use in the course CH462 Biochemistry II taught by R. Jeremy Johnson at the Butler University, Indianapolis, USA. This reservation includes Sandbox Reserved 1765 through Sandbox Reserved 1795. |
To get started:
More help: Help:Editing |
| |||||||||||
References
- ↑ 1.0 1.1 1.2 Yen PM. Physiological and molecular basis of thyroid hormone action. Physiol Rev. 2001 Jul;81(3):1097-142. doi: 10.1152/physrev.2001.81.3.1097. PMID: 11427693.
- ↑ Pirahanchi Y, Toro F, Jialal I. Physiology, Thyroid Stimulating Hormone. [Updated 2022 May 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK499850/
- ↑ 3.0 3.1 3.2 3.3 Duan J, Xu P, Luan X, Ji Y, He X, Song N, Yuan Q, Jin Y, Cheng X, Jiang H, Zheng J, Zhang S, Jiang Y, Xu HE. Hormone- and antibody-mediated activation of the thyrotropin receptor. Nature. 2022 Aug 8. pii: 10.1038/s41586-022-05173-3. doi:, 10.1038/s41586-022-05173-3. PMID:35940204 doi:http://dx.doi.org/10.1038/s41586-022-05173-3
- ↑ Kohn LD, Shimura H, Shimura Y, Hidaka A, Giuliani C, Napolitano G, Ohmori M, Laglia G, Saji M. The thyrotropin receptor. Vitam Horm. 1995;50:287-384. doi: 10.1016/s0083-6729(08)60658-5. PMID: 7709602.
- ↑ 5.0 5.1 5.2 Kleinau, G., Worth, C. L., Kreuchwig, A., Biebermann, H., Marcinkowski, P., Scheerer, P., & Krause, G. (2017). Structural–functional features of the thyrotropin receptor: A class A G-protein-coupled receptor at work. Frontiers in Endocrinology, 8. https://doi.org/10.3389/fendo.2017.00086
- ↑ 6.0 6.1 6.2 6.3 6.4 Faust B, Billesbolle CB, Suomivuori CM, Singh I, Zhang K, Hoppe N, Pinto AFM, Diedrich JK, Muftuoglu Y, Szkudlinski MW, Saghatelian A, Dror RO, Cheng Y, Manglik A. Autoantibody mimicry of hormone action at the thyrotropin receptor. Nature. 2022 Aug 8. pii: 10.1038/s41586-022-05159-1. doi:, 10.1038/s41586-022-05159-1. PMID:35940205 doi:http://dx.doi.org/10.1038/s41586-022-05159-1
- ↑ Yumiko Mizutori, Chun-Rong Chen, Sandra M. McLachlan, Basil Rapoport, The Thyrotropin Receptor Hinge Region Is Not Simply a Scaffold for the Leucine-Rich Domain but Contributes to Ligand Binding and Signal Transduction, Molecular Endocrinology, Volume 22, Issue 5, 1 May 2008, Pages 1171–1182, https://doi.org/10.1210/me.2007-0407
- ↑ 8.0 8.1 8.2 8.3 Faust, B., Billesbølle, C.B., Suomivuori, CM. et al. Autoantibody mimicry of hormone action at the thyrotropin receptor. Nature 609, 846–853 (2022). https://doi.org/10.1038/s41586-022-
- ↑ Virginie Vlaeminck-Guillem, Su-Chin Ho, Patrice Rodien, Gilbert Vassart, Sabine Costagliola, Activation of the cAMP Pathway by the TSH Receptor Involves Switching of the Ectodomain from a Tethered Inverse Agonist to an Agonist, Molecular Endocrinology, Volume 16, Issue 4, 1 April 2002, Pages 736–746, https://doi.org/10.1210/mend.16.4.0816
- ↑ Goricanec, D., Stehle, R., Egloff, P., Grigoriu, S., Plückthun, A., Wagner, G., & Hagn, F. (2016). Conformational dynamics of a G-protein α subunit is tightly regulated by nucleotide binding. Proceedings of the National Academy of Sciences, 113(26). https://doi.org/10.1073/pnas.1604125113
- ↑ Nunez Miguel R, Sanders J, Chirgadze DY, Furmaniak J, Rees Smith B. Thyroid stimulating autoantibody M22 mimics TSH binding to the TSH receptor leucine rich domain: a comparative structural study of protein-protein interactions. J Mol Endocrinol. 2009 May;42(5):381-95. Epub 2009 Feb 16. PMID:19221175 doi:10.1677/JME-08-0152
- ↑ 12.0 12.1 Chen, C.-R., McLachlan, S. M., & Rapoport, B. (2007). Suppression of thyrotropin receptor constitutive activity by a monoclonal antibody with inverse agonist activity. Endocrinology, 148(5), 2375–2382. https://doi.org/10.1210/en.2006-1754