This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Marcos Vinícius Caetano/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 14: | Line 14: | ||
*'''The tail domain''' (C-terminal): binds cargoes and anchors the protein to specific membrane compartments. | *'''The tail domain''' (C-terminal): binds cargoes and anchors the protein to specific membrane compartments. | ||
| - | '' | + | |
| + | '''''Important''': The entry <scene name='97/973101/Complete_structure_myovi/1'>'2BKI'</scene> contains only the motor domain and neck region of myosin VI. However, the entry '2KIA' crystallized Myosin VI C-terminal cargo-binding domain (tail domain).'' | ||
| Line 36: | Line 37: | ||
== Structure features== | == Structure features== | ||
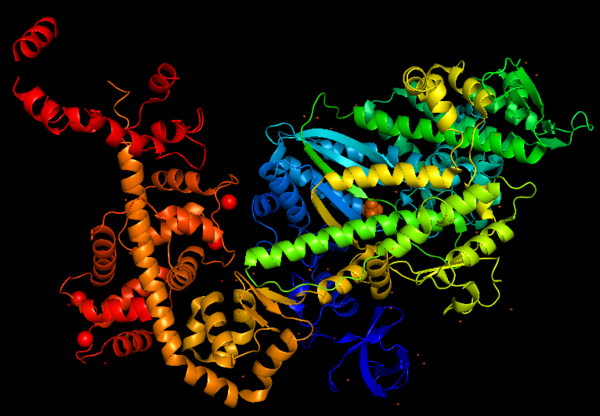
| - | The crystal structure of the entry '2BKI' was determined by | + | The crystal structure of the entry '2BKI' was determined by '''x-ray diffraction''' with the resolution of 2,90 Å and is composed by '''3 chains''': <scene name='97/973101/Myo_vi_chain_a/1'>Myosin VI</scene> and two chains of <scene name='97/973101/Chains_b_d/1'>Calmodulin</scene> (a protein that plays a major role in the Ca<sup>2+</sup>-dependent regulation of wide variety of cellular events). |
| Line 45: | Line 46: | ||
[[Image:MyoVI alpha and beta v2.png|600px]] | [[Image:MyoVI alpha and beta v2.png|600px]] | ||
''Figure 3: Representation of secondary structures of Myosin VI, alpha helix are shown in <span style="color:red">'''red'''</span> and β-sheet in <span style="color:gold">'''yellow'''</span>.'' | ''Figure 3: Representation of secondary structures of Myosin VI, alpha helix are shown in <span style="color:red">'''red'''</span> and β-sheet in <span style="color:gold">'''yellow'''</span>.'' | ||
| + | |||
'''Unique features''' | '''Unique features''' | ||
| Line 73: | Line 75: | ||
*'''Function''': Redirectionare the lever arm and contains a new CaM-binding motif. | *'''Function''': Redirectionare the lever arm and contains a new CaM-binding motif. | ||
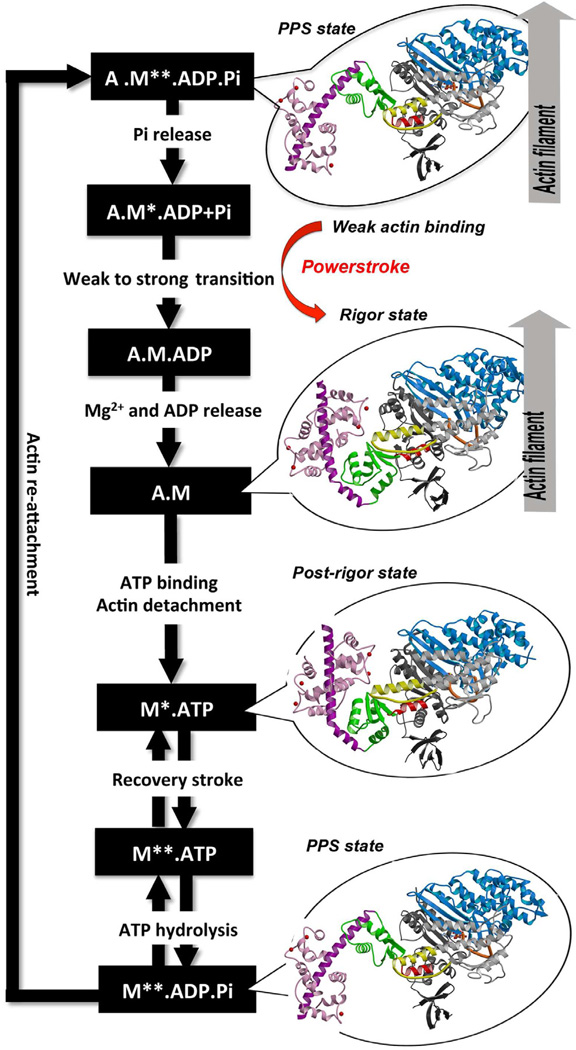
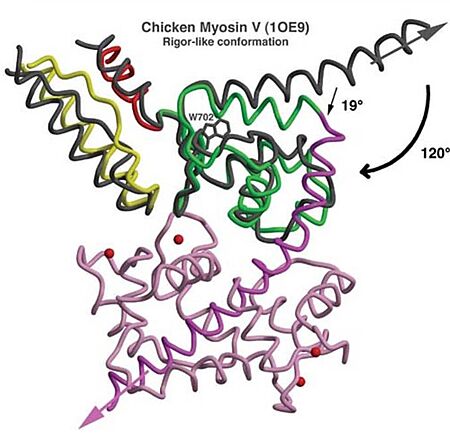
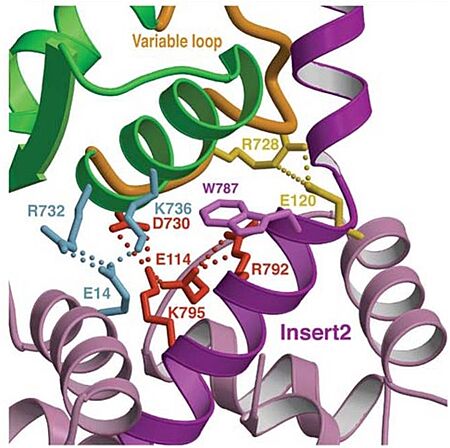
| - | *'''Mechanism''': The proximal part of insert 2 (<scene name='97/973101/774_812_distal_and_proximal/3'>Pro774-Trp787</scene> - in <span style="color:red">'''red'''</span>) wraps around the <scene name='97/973101/774_812_distal_and_proximal/4'>converter</scene> (in <span style="color:blue">'''blue'''</span>), while the distal part (<scene name='97/973101/774_812_distal_and_proximal/3'>Trp787-Tyr812</scene> - in <span style="color:green">'''green'''</span>) forms a CaM-binding motif. The insert 2 and its associated CaM molecule (with 4Ca<sup>2+</sup>), make specific interactions with the converter, many involving a variable loop (<scene name='97/973101/Insert_2_full/1'>Lys719-Pro731</scene>- in <span style="color:magenta">'''magenta'''</span>). In addiction, there is ''' | + | *'''Mechanism''': The proximal part of insert 2 (<scene name='97/973101/774_812_distal_and_proximal/3'>Pro774-Trp787</scene> - in <span style="color:red">'''red'''</span>) wraps around the <scene name='97/973101/774_812_distal_and_proximal/4'>converter</scene> (in <span style="color:blue">'''blue'''</span>), while the distal part (<scene name='97/973101/774_812_distal_and_proximal/3'>Trp787-Tyr812</scene> - in <span style="color:green">'''green'''</span>) forms a CaM-binding motif. The insert 2 and its associated CaM molecule (with 4Ca<sup>2+</sup>), make specific interactions with the converter, many involving a variable loop (<scene name='97/973101/Insert_2_full/1'>Lys719-Pro731</scene>- in <span style="color:magenta">'''magenta'''</span>). In addiction, there is '''apolar interactions''' that stabilize the proximal part of insert 2 on the surface of the converter, where <scene name='97/973101/Insert_2_full/3'>hidrophofobic side chains</scene> from the amino acids <span style="color:gold">'''F763, F766, M770'''</span> stabilize the orientation of the last helix <scene name='97/973101/Insert_2_full/4'>(representation with cartoon and stick)</scene>. The result of <scene name='97/973101/Insert_2_full/2'>interactions</scene> is that <scene name='97/973101/Iq_helix_emerge/1'>IQ helix</scene> - in <span style="color:green">'''green'''</span>) '''emerges ~120°''' from the position that it emerges in all other myosins, '''redirecting the IQ helix''' and the CaM towards the '''minus-end''' of the actin filament. The figure below compare the orientation of this IQ helix in myosin VI (green) with myosin V (black), and there is a difference of 19°, therefore, '''this is what makes myosin VI unique'''. |
[[Image:Comparison MyoVI and MyoV.jpg|450px]] | [[Image:Comparison MyoVI and MyoV.jpg|450px]] | ||
Revision as of 19:25, 11 July 2023
Myosin VI nucleotide-free (MDinsert2-IQ) crystal structure
| |||||||||||