This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Marcos Vinícius Caetano/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 26: | Line 26: | ||
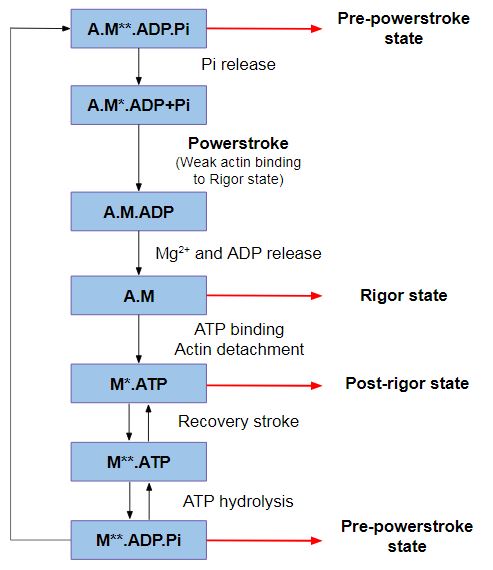
Despite the fact that myosin VI directionality is reversed, it has the '''same kinetic ATPase cycle''' of interaction with actin, as shown in the figure below. There are 3 states of different conformation: Pre-powerstroke, Rigor state and Post-rigor state <ref>Ménétrey, J., Isabet, T., Ropars, V., Mukherjea, M., Pylypenko, O., Liu, X., Perez, J., Vachette, P., Sweeney, H. L., & Houdusse, A. M. (2012). Processive steps in the reverse direction require uncoupling of the lead head lever arm of myosin VI. Molecular cell, 48(1), 75–86. https://doi.org/10.1016/j.molcel.2012.07.034</ref>. | Despite the fact that myosin VI directionality is reversed, it has the '''same kinetic ATPase cycle''' of interaction with actin, as shown in the figure below. There are 3 states of different conformation: Pre-powerstroke, Rigor state and Post-rigor state <ref>Ménétrey, J., Isabet, T., Ropars, V., Mukherjea, M., Pylypenko, O., Liu, X., Perez, J., Vachette, P., Sweeney, H. L., & Houdusse, A. M. (2012). Processive steps in the reverse direction require uncoupling of the lead head lever arm of myosin VI. Molecular cell, 48(1), 75–86. https://doi.org/10.1016/j.molcel.2012.07.034</ref>. | ||
| - | [[Image: | + | [[Image:Cycle ATP.JPG|500px]] |
| - | + | ''Figure 2: ATPase cycle of myosins.'' | |
| - | + | ||
'''Pre-powerstroke''': myosin binds to actin, it has hydrolyzed ATP, but keeps the products MgADP and Pi. The interaction of myosin with actin results in the release of Pi, which increases the binding affinity for actin and triggers the release of MgADP to form a rigor conformation on actin. At this moment, occurs the powerstroke, a large movement (11 nm) of the lever arm between the pre-powerstroke and rigor state, during which the converter and the CaM rotates and alters its conformation. | '''Pre-powerstroke''': myosin binds to actin, it has hydrolyzed ATP, but keeps the products MgADP and Pi. The interaction of myosin with actin results in the release of Pi, which increases the binding affinity for actin and triggers the release of MgADP to form a rigor conformation on actin. At this moment, occurs the powerstroke, a large movement (11 nm) of the lever arm between the pre-powerstroke and rigor state, during which the converter and the CaM rotates and alters its conformation. | ||
| Line 36: | Line 35: | ||
'''Post-rigor state''': Once the myosin has dissociated, there is a rapid and reversible isomerization between the post-rigor state, which cannot hydrolyze ATP, and the pre-powerstroke state that can rapidly hydrolyze ATP due to repositioning of a nucleotide-binding switch II, whose conformational change in the motor that repositions the myosin lever arm, repriming the lever arm for movement on actin. | '''Post-rigor state''': Once the myosin has dissociated, there is a rapid and reversible isomerization between the post-rigor state, which cannot hydrolyze ATP, and the pre-powerstroke state that can rapidly hydrolyze ATP due to repositioning of a nucleotide-binding switch II, whose conformational change in the motor that repositions the myosin lever arm, repriming the lever arm for movement on actin. | ||
| - | == | + | == Structural features== |
All data described below was based in the article from Ménétrey et. al, 2012 <ref>Ménétrey, J., Isabet, T., Ropars, V., Mukherjea, M., Pylypenko, O., Liu, X., Perez, J., Vachette, P., Sweeney, H. L., & Houdusse, A. M. (2012). Processive steps in the reverse direction require uncoupling of the lead head lever arm of myosin VI. Molecular cell, 48(1), 75–86. https://doi.org/10.1016/j.molcel.2012.07.034</ref>. | All data described below was based in the article from Ménétrey et. al, 2012 <ref>Ménétrey, J., Isabet, T., Ropars, V., Mukherjea, M., Pylypenko, O., Liu, X., Perez, J., Vachette, P., Sweeney, H. L., & Houdusse, A. M. (2012). Processive steps in the reverse direction require uncoupling of the lead head lever arm of myosin VI. Molecular cell, 48(1), 75–86. https://doi.org/10.1016/j.molcel.2012.07.034</ref>. | ||
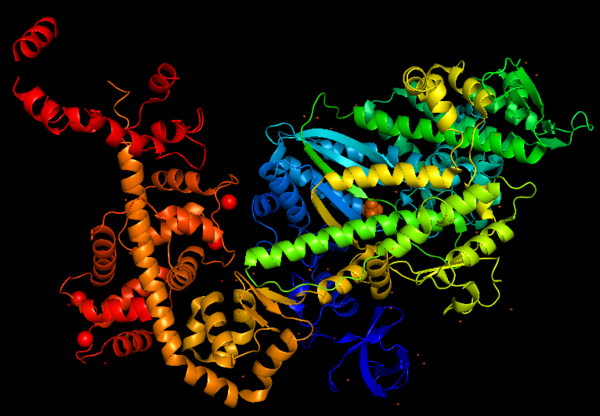
The crystal structure of the entry '2BKI' was determined by '''x-ray diffraction''' with the resolution of 2,90 Å and is composed by '''3 chains''': <scene name='97/973101/Myo_vi_chain_a/1'>Myosin VI</scene> and two chains of <scene name='97/973101/Chains_b_d/1'>Calmodulin</scene> (a protein that plays a major role in the Ca<sup>2+</sup>-dependent regulation of wide variety of cellular events). | The crystal structure of the entry '2BKI' was determined by '''x-ray diffraction''' with the resolution of 2,90 Å and is composed by '''3 chains''': <scene name='97/973101/Myo_vi_chain_a/1'>Myosin VI</scene> and two chains of <scene name='97/973101/Chains_b_d/1'>Calmodulin</scene> (a protein that plays a major role in the Ca<sup>2+</sup>-dependent regulation of wide variety of cellular events). | ||
Revision as of 20:59, 14 July 2023
Myosin VI nucleotide-free (MDinsert2-IQ) crystal structure (2BKI)
| |||||||||||