This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1qr0
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
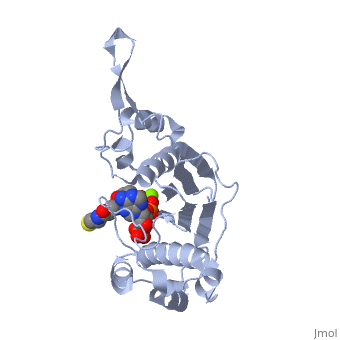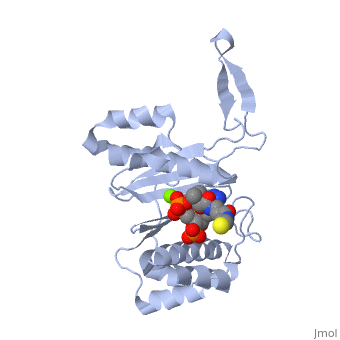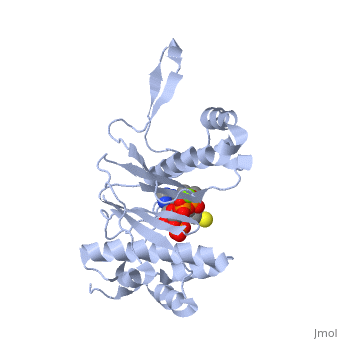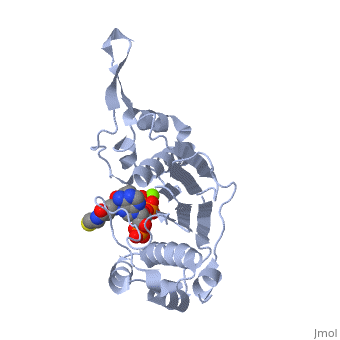
<StructureSection load='1qr0' size='340' side='right'caption='[[1qr0]], [[Resolution|resolution]] 1.90Å' scene=''> | <StructureSection load='1qr0' size='340' side='right'caption='[[1qr0]], [[Resolution|resolution]] 1.90Å' scene=''> | ||
== Structural highlights == | == Structural highlights == | ||
| - | <table><tr><td colspan='2'>[[1qr0]] is a 1 chain structure with sequence from [ | + | <table><tr><td colspan='2'>[[1qr0]] is a 1 chain structure with sequence from [https://en.wikipedia.org/wiki/Bacillus_subtilis Bacillus subtilis]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1QR0 OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=1QR0 FirstGlance]. <br> |
| - | </td></tr><tr id=' | + | </td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 1.9Å</td></tr> |
| - | <tr id=' | + | <tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=COA:COENZYME+A'>COA</scene>, <scene name='pdbligand=MG:MAGNESIUM+ION'>MG</scene></td></tr> |
| - | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[ | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=1qr0 FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=1qr0 OCA], [https://pdbe.org/1qr0 PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=1qr0 RCSB], [https://www.ebi.ac.uk/pdbsum/1qr0 PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=1qr0 ProSAT]</span></td></tr> |
</table> | </table> | ||
== Function == | == Function == | ||
| - | [ | + | [https://www.uniprot.org/uniprot/SFP_BACSU SFP_BACSU] Activates the seven peptidyl carrier protein (PCP) domains of surfactin synthase SRF1/2/3 by transferring the 4'-phosphopantetheinyl moiety of coenzyme A (CoA) to a serine residue. Required for cells of B.subtilis to become producers of the lipopeptide antibiotics surfactin and plipastatin B1.<ref>PMID:8939709</ref> |
== Evolutionary Conservation == | == Evolutionary Conservation == | ||
[[Image:Consurf_key_small.gif|200px|right]] | [[Image:Consurf_key_small.gif|200px|right]] | ||
| Line 20: | Line 20: | ||
</jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/main_output.php?pdb_ID=1qr0 ConSurf]. | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/main_output.php?pdb_ID=1qr0 ConSurf]. | ||
<div style="clear:both"></div> | <div style="clear:both"></div> | ||
| - | <div style="background-color:#fffaf0;"> | ||
| - | == Publication Abstract from PubMed == | ||
| - | The Bacillus subtilis Sfp protein activates the peptidyl carrier protein (PCP) domains of surfactin synthetase by transferring the 4'-phosphopantetheinyl moiety of coenzyme A (CoA) to a serine residue conserved in all PCPs. Its wide PCP substrate spectrum renders Sfp a biotechnologically valuable enzyme for use in combinatorial non-ribosomal peptide synthesis. The structure of the Sfp-CoA complex determined at 1.8 A resolution reveals a novel alpha/beta-fold exhibiting an unexpected intramolecular 2-fold pseudosymmetry. This suggests a similar fold and dimerization mode for the homodimeric phosphopantetheinyl transferases such as acyl carrier protein synthase. The active site of Sfp accommodates a magnesium ion, which is complexed by the CoA pyrophosphate, the side chains of three acidic amino acids and one water molecule. CoA is bound in a fashion that differs in many aspects from all known CoA-protein complex structures. The structure reveals regions likely to be involved in the interaction with the PCP substrate. | ||
| - | |||
| - | Crystal structure of the surfactin synthetase-activating enzyme sfp: a prototype of the 4'-phosphopantetheinyl transferase superfamily.,Reuter K, Mofid MR, Marahiel MA, Ficner R EMBO J. 1999 Dec 1;18(23):6823-31. PMID:10581256<ref>PMID:10581256</ref> | ||
| - | |||
| - | From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | ||
| - | </div> | ||
| - | <div class="pdbe-citations 1qr0" style="background-color:#fffaf0;"></div> | ||
== References == | == References == | ||
<references/> | <references/> | ||
__TOC__ | __TOC__ | ||
</StructureSection> | </StructureSection> | ||
| - | [[Category: | + | [[Category: Bacillus subtilis]] |
[[Category: Large Structures]] | [[Category: Large Structures]] | ||
| - | [[Category: Ficner | + | [[Category: Ficner R]] |
| - | [[Category: Marahiel | + | [[Category: Marahiel AM]] |
| - | [[Category: Mofid | + | [[Category: Mofid RM]] |
| - | [[Category: Reuter | + | [[Category: Reuter K]] |
| - | + | ||
| - | + | ||
Current revision
CRYSTAL STRUCTURE OF THE 4'-PHOSPHOPANTETHEINYL TRANSFERASE SFP-COENZYME A COMPLEX
| |||||||||||