Antizyme Inhibitor
From Proteopedia
| Line 23: | Line 23: | ||
binding suggest that AzI does not bind PLP. A comparison to the <scene name='3btn/Plp/1'>PLP binding site of ODC</scene> revealed that AzI lacks the residues participating in PLP binding. Biochemical studies confirmed the lack of PLP binding and revealed that AzI exists as a monomer in solution while ODC is dimeric. Our findings that AzI exists as a monomer and its inability to bind PLP provide two independent explanations for its lack of enzymatic activity, and suggest the basis for its enhanced affinity towards Az. | binding suggest that AzI does not bind PLP. A comparison to the <scene name='3btn/Plp/1'>PLP binding site of ODC</scene> revealed that AzI lacks the residues participating in PLP binding. Biochemical studies confirmed the lack of PLP binding and revealed that AzI exists as a monomer in solution while ODC is dimeric. Our findings that AzI exists as a monomer and its inability to bind PLP provide two independent explanations for its lack of enzymatic activity, and suggest the basis for its enhanced affinity towards Az. | ||
{{Clear}} | {{Clear}} | ||
| - | applet load="AzIODCali.pdb" size="500" color="white" frame="true" align="right" spinBox="true" /> | + | <applet load="AzIODCali.pdb" size="500" color="white" frame="true" align="right" spinBox="true" /> |
'''Comparison of the AzI structure to that of ODC''' | '''Comparison of the AzI structure to that of ODC''' | ||
Revision as of 13:01, 6 July 2008
Crystal structure of the Antizyme inhibitor
| |||||||
| AzI, unpublished structure | |||||||
|---|---|---|---|---|---|---|---|
| Coordinates: | save as pdb, mmCIF, xml | ||||||
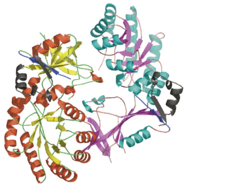
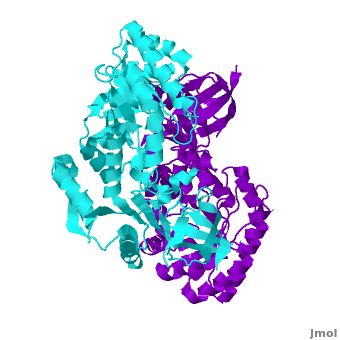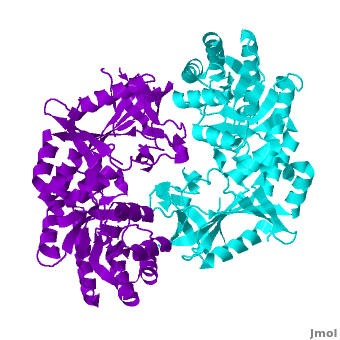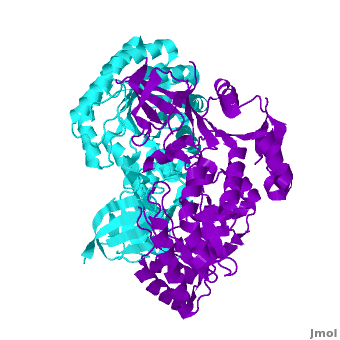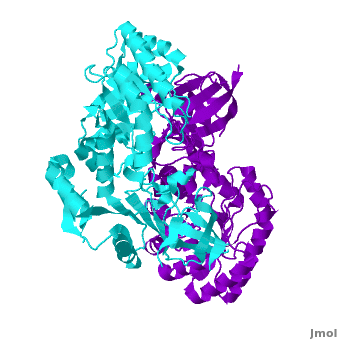
Antizyme inhibitor (AzI) regulates cellular polyamine homeostasis by binding to the polyamine-induced protein, Antizyme (Az), with greater affinity than ODC. AzI is highly homologous to ornithine decarboxylase (ODC), but is not enzymatically active. In order to understand these specific characteristics of AzI and its differences from ODC, we determined the 3D structure of mouse AzI to 2.05Å resolution. Both AzI and ODC crystallize as a (one monomer in blue and the other in blue violet). However, fewer interactions at the dimer , a smaller buried surface area, and lack of symmetry of the interactions between residues from the two monomers in the AzI structure suggest that this dimeric structure is non-physiological. In addition, the absence of residues and interactions required for binding suggest that AzI does not bind PLP. A comparison to the revealed that AzI lacks the residues participating in PLP binding. Biochemical studies confirmed the lack of PLP binding and revealed that AzI exists as a monomer in solution while ODC is dimeric. Our findings that AzI exists as a monomer and its inability to bind PLP provide two independent explanations for its lack of enzymatic activity, and suggest the basis for its enhanced affinity towards Az.
|
Comparison of the AzI structure to that of ODC
Primary Reference
Shira Albeck, Orly Dym, Tamar Unger, Zohar Snapir, Zippy Bercovich and Chaim Kahana. Crystallographic and biochemical studies revealing the structural basis for antizyme inhibitor function. Protein Sci. 2008 May; 17(5): 793-802. Epub 2008 Mar 27.
Proteopedia Page Contributors and Editors (what is this?)
Alexander Berchansky, Michal Harel, Joel L. Sussman, Dinesh Kulhary, David Canner, Jaime Prilusky, Orly Dym