This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Aconitase
From Proteopedia
| Line 5: | Line 5: | ||
In most organims, there is a cytosolic enzyme with an ACO domain (cAc), and in eukaryotes, a second copy of it was introduced with mitochondria (mAc). Plants developed even more copies in mitochondria. | In most organims, there is a cytosolic enzyme with an ACO domain (cAc), and in eukaryotes, a second copy of it was introduced with mitochondria (mAc). Plants developed even more copies in mitochondria. | ||
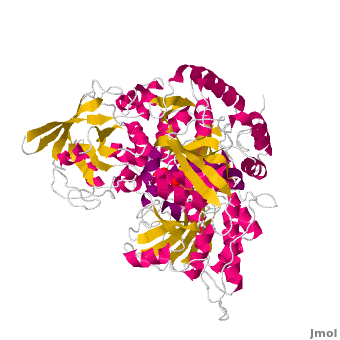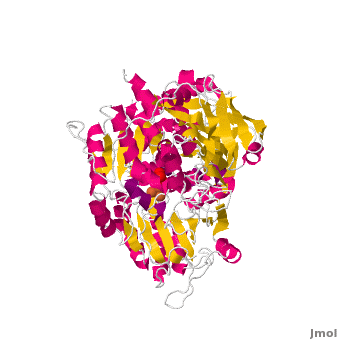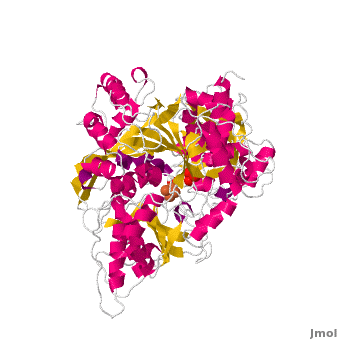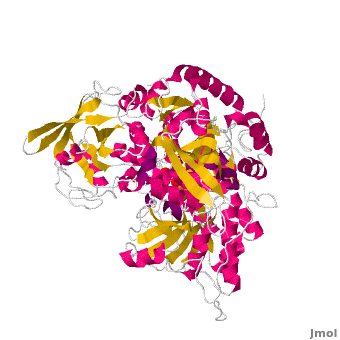
| - | <applet load='Morph_2ipy-2b3x.pdb.gz' scene='Aconitase/ | + | <applet load='Morph_2ipy-2b3x.pdb.gz' scene='Aconitase/2ipy-total/2' size='400' frame='true' align='right' caption="" />A specialty of cAc is that in mammals it has developed a <scene name='Aconitase/2ipy-total/2'>second function</scene> as inhibitor of <scene name='Aconitase/2ipy-rna/1'>those mRNA</scene> that carry an <scene name='Aconitase/2ipy-rna-ire/1'>iron-regulatory element (IRE)</scene>. Therefore, the cytosolic cAc is named IREBP for IRE-binding protein when this function is talked about. Only one of the two functions is active, depending on whether the (4Fe4S) cofactor is present in the molecule: it's essential for the ACO function. You can see, by <scene name='Aconitase/Morph/1'>looking at the morph</scene>, how much the enzyme structure differs between those two functions. |
<!--== Available structures == | <!--== Available structures == | ||
Revision as of 10:25, 18 February 2009
Aconitase (ACO) is an enzymatic domain that confers the ability to catalyse the equilibrium
- citrate = aconitate + H2O = isocitrate
This reaction is part of the citrate (TCA-, Krebs-)cycle.
In most organims, there is a cytosolic enzyme with an ACO domain (cAc), and in eukaryotes, a second copy of it was introduced with mitochondria (mAc). Plants developed even more copies in mitochondria.
|
Weblinks
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Ralf Stephan, David Canner, Joel L. Sussman, Jaime Prilusky, Anthony Noles, Angel Herraez, Eran Hodis