This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
YbgC
From Proteopedia
| |||||||||
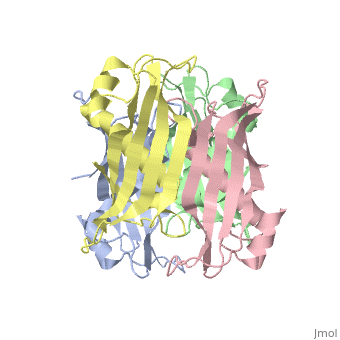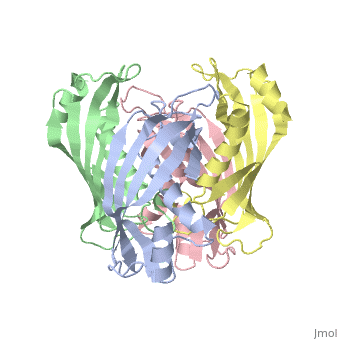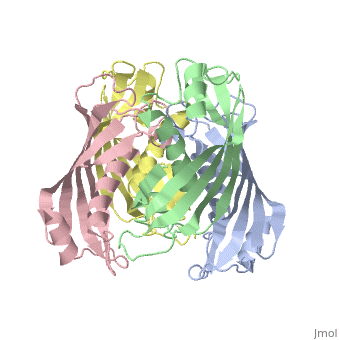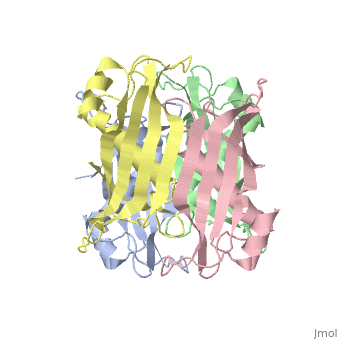
| 2pzh, resolution 1.70Å () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Gene: | HP0496 (Helicobacter pylori) | ||||||||
| Related: | 1lo7, 1s5u | ||||||||
| |||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, RCSB, PDBsum | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
YbgCis believed to have first appeared in the Tol complex with the separation of the δε proteobacteria from the αβγ proteobacteria[1].
Structure
YbgC is a cytoplasmic protein in the Tol-Pal complex[2]. In Helicobacter pylori, the protein is part of a 'hot-dog' family of proteins, with an epsilongamma tetrameric arrangement[3].
Function
The protein displays thioesterase activity towards acyl-CoA thioesters[4], and has a strong sequence conservation with its active site residues with other proteins of a similar function[5] for example with the Pseudonomas sp. strain CBS3 4-hydroxybenzoyl-CoA thioesterase[6].
However, its role within the Tol complex itself remains unknown, although it may be involved in the acetylation of another protein within the complex, with the role of an activity-regulator[1]. It is also thought to be involved in the cell division complex of Tol-Pal[7].
References
- ↑ 1.0 1.1 Sturgis JN. Organisation and evolution of the tol-pal gene cluster. J Mol Microbiol Biotechnol. 2001 Jan;3(1):113-22. PMID:11200223
- ↑ Walburger A, Lazdunski C, Corda Y. The Tol/Pal system function requires an interaction between the C-terminal domain of TolA and the N-terminal domain of TolB. Mol Microbiol. 2002 May;44(3):695-708. PMID:11994151
- ↑ Angelini A, Cendron L, Goncalves S, Zanotti G, Terradot L. Structural and enzymatic characterization of HP0496, a YbgC thioesterase from Helicobacter pylori. Proteins. 2008 Mar 12;72(4):1212-1221. PMID:18338382 doi:http://dx.doi.org/10.1002/prot.22014
- ↑ Angelini A, Cendron L, Goncalves S, Zanotti G, Terradot L. Structural and enzymatic characterization of HP0496, a YbgC thioesterase from Helicobacter pylori. Proteins. 2008 Mar 12;72(4):1212-1221. PMID:18338382 doi:http://dx.doi.org/10.1002/prot.22014
- ↑ Benning MM, Wesenberg G, Liu R, Taylor KL, Dunaway-Mariano D, Holden HM. The three-dimensional structure of 4-hydroxybenzoyl-CoA thioesterase from Pseudomonas sp. Strain CBS-3. J Biol Chem. 1998 Dec 11;273(50):33572-9. PMID:9837940
- ↑ Zhuang Z, Song F, Martin BM, Dunaway-Mariano D. The YbgC protein encoded by the ybgC gene of the tol-pal gene cluster of Haemophilus influenzae catalyzes acyl-coenzyme A thioester hydrolysis. FEBS Lett. 2002 Apr 10;516(1-3):161-3. PMID:11959124
- ↑ Angelini A, Cendron L, Goncalves S, Zanotti G, Terradot L. Structural and enzymatic characterization of HP0496, a YbgC thioesterase from Helicobacter pylori. Proteins. 2008 Mar 12;72(4):1212-1221. PMID:18338382 doi:http://dx.doi.org/10.1002/prot.22014