This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
YbgF
From Proteopedia
Template:STRUCTURE 2xev Template:STRUCTURE 2wz7
Structure
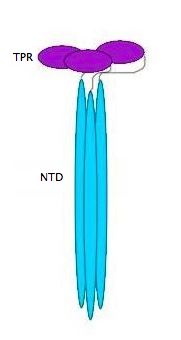
YbgF is a periplasmic protein with an N-terminal coiled coil domain (NTD) and a C-terminal tetratricopeptide domain (TPR), both of which are autonomous[1]. As seen in the 3D structure 2WZ7, the NTD forms an elongated trimer which is connected via a flexible linker to the TPR trimer, as seen in 2XEV. This connection can be cleaved by proteases[2].
Function
YbgF is a periplasmic protein which interacts with the C-terminal domain of TolA (TolAIII) during the import of colicin A into the cytoplasm[3].
References
- ↑ 1.0 1.1 Gerding MA, Ogata Y, Pecora ND, Niki H, de Boer PA. The trans-envelope Tol-Pal complex is part of the cell division machinery and required for proper outer-membrane invagination during cell constriction in E. coli. Mol Microbiol. 2007 Feb;63(4):1008-25. PMID:17233825 doi:10.1111/j.1365-2958.2006.05571.x
- ↑ Krachler AM, Sharma A, Cauldwell A, Papadakos G, Kleanthous C. TolA modulates the oligomeric status of YbgF in the bacterial periplasm. J Mol Biol. 2010 Oct 22;403(2):270-85. Epub 2010 Sep 15. PMID:20816983 doi:10.1016/j.jmb.2010.08.050
- ↑ Walburger A, Lazdunski C, Corda Y. The Tol/Pal system function requires an interaction between the C-terminal domain of TolA and the N-terminal domain of TolB. Mol Microbiol. 2002 May;44(3):695-708. PMID:11994151
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Laura McCauley, Joel L. Sussman, Alexander Berchansky
