This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Group:MUZIC:DARP
From Proteopedia

| |||||||||
| 1n0r, resolution 1.50Å () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ligands: | |||||||||
| Related: | 1n0q | ||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, PDBsum, RCSB | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
Introduction
Diabetes related ankyrin repeat protein DARP (Ankrd23) and its two close homologs Ankrd2/Arpp and Ankrd1/CARP correspond to a conserved gene family of muscle ankyrin repeat proteins (MARPs). [1] DARP is expressed in both heart and skeletal muscle (in addition to brown fat) and was identified by its upregulation in Type 2 diabetes and insulin-resistant animals, suggesting a potential role in energy metabolism. [2]
Contents |
Structure
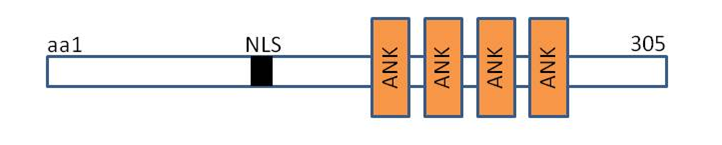
DARP contains putative nuclear localization signals,four tandem ankyrin-like repeats, potential coiled-coil dimerization motif within its unique aminoterminal domain that mediates the formation of homodimers. [3]The amino acid sequence of DARP showed high similarity to cardiac ankyrin-repeat protein (CARP) and ankyrin-repeat domain 2 (Ankrd2) with 45 and 36% identities, respectively
Gene Function
DARP knock out muscle fibers were less stiff, tended to have longer resting sarcomere lengths, and expressed a longer isoform of titin than their wild-type counterparts, indicating that this protein may play a role in the passive mechanical behavior of muscle. [4] DARP expression is altered by a change of energy supply and energy metabolic condition, induced by excess fatty acid treatment in vitro and fasting in vivo. [2] The expression of DARP is induced during recovery following starvation.
Interaction
DARP interacts with a tyrosine-rich binding motif between Ig80 and Ig81 of titin and with myopalladin. [1]
Refrences
- ↑ 1.0 1.1 Miller MK, Bang ML, Witt CC, Labeit D, Trombitas C, Watanabe K, Granzier H, McElhinny AS, Gregorio CC, Labeit S. The muscle ankyrin repeat proteins: CARP, ankrd2/Arpp and DARP as a family of titin filament-based stress response molecules. J Mol Biol. 2003 Nov 7;333(5):951-64. PMID:14583192
- ↑ 2.0 2.1 Ikeda K, Emoto N, Matsuo M, Yokoyama M. Molecular identification and characterization of a novel nuclear protein whose expression is up-regulated in insulin-resistant animals. J Biol Chem. 2003 Feb 7;278(6):3514-20. Epub 2002 Nov 26. PMID:12456686 doi:10.1074/jbc.M204563200
- ↑ Witt SH, Labeit D, Granzier H, Labeit S, Witt CC. Dimerization of the cardiac ankyrin protein CARP: implications for MARP titin-based signaling. J Muscle Res Cell Motil. 2005;26(6-8):401-8. PMID:16450059 doi:10.1007/s10974-005-9022-9
- ↑ Barash IA, Bang ML, Mathew L, Greaser ML, Chen J, Lieber RL. Structural and regulatory roles of muscle ankyrin repeat protein family in skeletal muscle. Am J Physiol Cell Physiol. 2007 Jul;293(1):C218-27. Epub 2007 Mar 28. PMID:17392382 doi:10.1152/ajpcell.00055.2007