This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sigma factor
From Proteopedia
OverviewSigma (σ) factor is the peoptide subunit needed for the initiation of RNA transcription in prokaryotic organisms . As opposed to eukaryotes, who utilize a variety of proteins to initiate gene transcription, prokaryotic transcription is initiated almost completely by a σ-factor. The large and biologically essential protein, RNA polymerase (RNAP), contains one σ-subunit, which binds , located upstream of transcription start sites. Function and StructureThe σ-factor performs two chief functions: to direct the catalytic core of RNAP to the promotoer upstream of the +1 start site of transcription, and finally to assist in the initiation of strand seperation of double-helical DNA, forming the transcription "bubble."[1] Each gene promoter utilizes a specific promoter region about 40 bp upstream of the transcription start site, and therefore different σ-factors play a role in the regulation of different genes [2]. This process, which includes association of the σ-factor with RNAP to recognize and open DNA at the promoter site, followed by dissociation of the σ to allow elongation, which can then activate additional RNAP enzymes, is referred to as the σ-cycle [3]. Domain Strucure & DNA interactionsThere are many types of σ-subunits, and each recognizes a unique promoter sequence. Furthmore, each unique σ is composed of a variable number of structured domains. The simplest σ-factors have two domains, few have three, and most, called housekeeping σ-factors, have 4 domains, given the names σ(4), σ(3), σ(2), and σ(1.1) [1,3]. All domains are linked by very flexible peptide linkers which can extend very long distances. Each of these domains utilizes DNA-binding determinants, or domains that recognize specific sequences and conformations in DNA. Most commonly, these recognized sequences occur at the -35 and -10 locations upstream of the +1 site. One such DNA-binding motif, the helix-turn-helix motif (), helps specifically recognize DNA promoters at both the -35 and -10 positions [1]. This HTH motif, used by most σ-factors, maintains its specificity and accuracy by binding in the major groove of DNA, where it can interact with the base pairs in the DNA double-helix. In many prokaryotes, these portions of DNA maintain consensus adenosine and thymine sequences [1,2], such as . Transcription BubbleThe , also referred to as the open complex is formed through the common housekeeping σ factors which unwind about 13 bp of duplex DNA in an ATP independent process. Research has shown that σ factors require invariant basic and aromatic residues (Phe, Tyr, Trp) critical for this formation [1]. The process of bubble formation begins at the -11 formation (usually A) and propogates to +1 site, through a phenomenon called , which interrupts the stacking interactions stabilizing the double helix conformation [1]. As this process occurs and the DNA transitions into the open promoter complex, certain RNAP-σ contacts are lost, initiating the dissociation of σ. In summary, the processes of -35 and -10 motif sequence recognition and helix strand separation are coupled by the σ factor. RestrictionInitiation of prokaryotic transcription requires cooperation between the σ peptide and RNAP. Without these fundamental interactions, no transcription is possible. Comformational and AutoinhibitoryNormally, σ-factor domains cannot bind to promoters on their own. These domains usually are placed in very compacted positions relative to each other, a conformation that buries DNA-binding determinants. This type of restriction is called conformational restriction[1]. Additionally, in housekeeping σs, a domain called the σ(1.1) stabilizes the compact conformation mentioned above, thereby preventing any promoter recognition. This method of restricting the binding abilities of isolated σ's is called autoinhibitory inhibition[1]. anti-σ'sAn additional method of restriction is through the action of anti-σ's, which act by making stable interactions with σ-domains, such as σ (4), which allows them to make energy-favorable interactions with RNAP residues. This causes a cascading "peeling off" effect of other σ-domains from the RNAP, preventing any interaction with duplex DNA and inhibiting transcription in an analogous process to competitive inhibition [3]. Gene Regulation and DifferentiationSince σ-factors are exclusively linked to gene expression in prokaryotic organisms, the variety of σ-factors in a cell dictate how and what genes are transcribed. Specialized function in cells, therefore, is highly moderated by its arsenal of σ-subunits. In fact, cellular development and differentiation are directly impacted and carried out by "cascades" of σ-factors. In the early stages of development, early genes[2] are transcribed by basic bacterial σ-factors. These genes are therefore transcribed to give new σ-factors, which in turn activate additional genes, and so on [2]. This process of σ-factor cascades demonstrates the versatile and essential biologic functions of the RNAP subunit, σ.
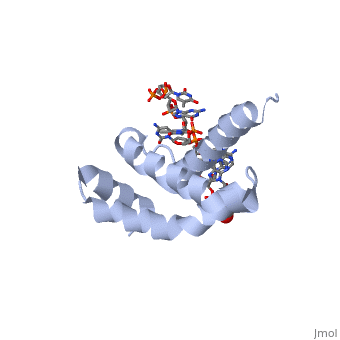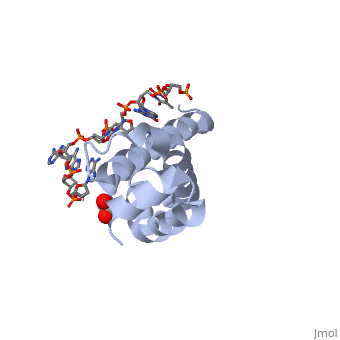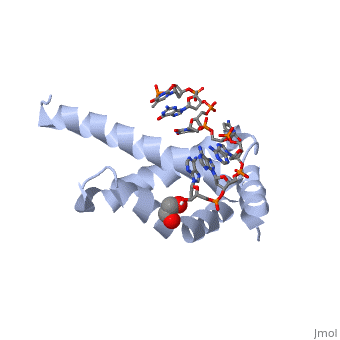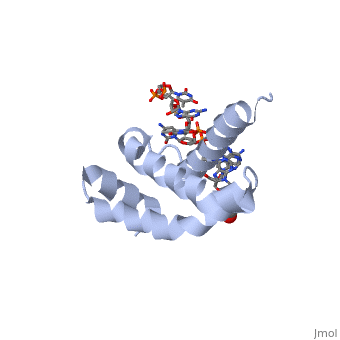
3D structures of sigma factorUpdated on 14-October-2014 Sigma factor1ku2 – TaSF region 1.2-3.1 – Thermus aquaticus Sigma factor complex with DNA3ugo, 3ugp - TaSF region 2 + DNA Sigma factor complex with protein3lev – TaSF residues 93-271 + antibody Sigma factor complex with protein and DNA1rio - TaSF region 4 + repression protein CI + DNA References1. Felklistov, Andrey, Brian D. Sharon, Seth A. Darst, and Carol A. Gross. "Bacterial Sigma Factors: A Historical, Structural, and Genomic Perspective." The Annual Review of Microbiology 68 (2014): 357-76. 2. Voet, Donald, Judith G. Voet, and Charlotte W. Pratt. Fundamentals of Biochemistry: Life at the Molecular Level. 3rd ed. Hoboken, NJ: Wiley, 2008. 3. Mooney, R. A., S. A. Darst, and R. Landick. "Sigma and RNA Polymerase: An On-again, Off-again Relationship?" Molecular Cell 20.3 (2005): 335-45.
| ||||||||||||