Sandbox Reserved 1627
From Proteopedia
Homo sapiens Insulin Receptor
IntroductionThe insulin receptor is a transmembrane protein receptor that is activated by the binding of insulin and is a vital proponent of cellular function. The insulin receptor belongs to the large class of tyrosine kinase receptors (RTKs). RTKs have a high affinity at the cell surface to bind to a particular ligand and are made up of three distinct parts: an extracellular domain with binding sites, a transmembrane region, and an intracellular domain with the tyrosine kinases that lead to downstream signaling upon activation of each other. The unique ability of the insulin receptor in particular to change conformation allows it to play a key role in a variety of cellular pathways including glucose homeostasis, regulation of lipid, protein, and carbohydrate metabolism, gene expression, and even modulation of brain neurotransmitter levels. This page focuses specifically on the insulin receptor's role in glucose homeostasis. Structural OverviewThe insulin receptor is a dimer of heterodimers made of two alpha subunits and two beta subunits [1].The are on the extracellular side of the membrane and are critical for binding insulin. The have the potential to interact with insulin ligands on the extracellular side of the membrane. There can be up to four binding sites, but it is generally more common for only one or two insulin molecules to bind to the receptor due to the occurrence of negative affinity at the binding site. The are transmembrane subunits that contain a tyrosine kinase region. When the entire receptor experiences a conformation change from the V shape to the T shape upon activation or binding of an insulin molecule, the Beta chains are brought in close proximity to each other. When the two subunits are brought near to each other in the activated T form, the Tyrosine Kinase regions are able to autophosphorylate their counterparts at particular Tyrosine locations. It is important to note in the overall discussion of the insulin receptor structure that it has only been imaged in pieces, and not as a whole at this point in time. There are proposed structures of the entire molecule based off of the known function of the autophosphorylation, but the structure discussed throughout this page only contains part of the Beta subunits and is supported by the works from Uchikawa E, et al.. Function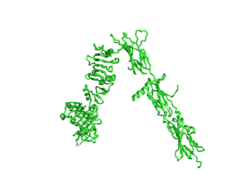 Figure 1. An image of the inactive Insulin Receptor Alpha subunit in the inverted V conformation. This image shows only a protomer of the inactive alpha subunit because the entire inactive alpha subunit dimer has been unable to be photographed because the transition state has yet to be determined PDB: 6CE7. 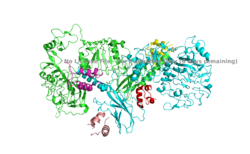 Figure 2. An image of the dimer of an active alpha subunit. The light blue and green areas represent the alpha subunit of the insulin receptor. The magenta, light pink, yellow, and red colorings are the four insulin binding sites with insulin bound. PDB: 6PXW. 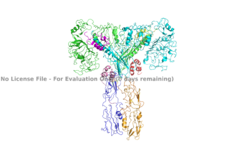 Figure 3. An image of the active insulin receptor showing both the alpha and beta subunits and insulin bound to the four binding sites. The dimers of the alpha subunit are colored in light blue and green. The intracellular beta sites are colored in blue and orange. The insulin binding sites with insulin bound are colored in magenta, light pink, yellow, and red. PDB: 6SOF. The insulin receptor's structure is critical to it's function. In regards to glucose homeostasis, the receptor begins the signaling pathway that will eventually move glucose transporters to the cell surface which will allow glucose to passively defuse into the cell. The glucose receptor is inactive in the absence of insulin. When insulin does bind to the receptor, it undergoes a conformation change from the inactive inverted V state (Figure 1) to the active T state (Figures 2 and 3). Once activated, the intracellular Beta subunits move together close enough to autophosphorylate, and downstream signaling begins by the phosphorylation of the Insulin Receptor Substrate (IRS). StructureThe insulin receptor is a receptor tyrosine kinase that exists in two stable conformations, an inactive and active state. The entire insulin receptor is a dimer of heterodimers with two extracellular alpha subunits, and two transmembrane/intracellular beta subunits linked and stabilized by multiple disulfide bonds. Alpha SubunitThe alpha subunit (Figures 1 & 2) is the extracellular domain of the insulin receptor and is the site of insulin binding. The alpha subunit is comprised of two Leucine rich domains (L1 & L2), a Cysteine rich domain (CR), and a C-terminal alpha helix. The actual site of insulin binding occurs at the and is stabilized by the L1 and L2 domains. Beta SubunitThe beta subunit is the membrane spanning and intracellular portion of the insulin receptor. This domain is composed of three Fibronectin domains (FN III-1,-2,-3) and the tyrosine kinase domain. The tyrosine kinase domain is the site of autophosphorylation upon activation of the receptor. Conformation ChangeThe inactive form of the insulin receptor predominates in low-levels of circulating insulin, whereas the active conformation is seen when insulin binds to any of the 4 receptor sites. The inactive conformation resembles an inverted V (Figure 1), and the active conformation resembles a T (Figure 3). Upon the binding of insulin to any of the four binding sites, the conformation change will begin, causing the Beta subunit's tyrosine kinase domains to move close together, allowing them to autophosphorylate. This autophosphorylation is what activates the insulin receptor and allows it to participate in further downstream signaling pathways. [2]. The insulin receptor can maximally bind four insulins to the active T-state at the four distinct insulin binding sites. Once insulin binds, the structural change from inactive to active state is stabilized by a tripartite interaction between insulin, L1, the C-terminal alpha helix, and the FNIII-1 domain. Studies have found that optimal insulin receptor activation requires the binding of multiple insulin ligands to two insulin binding sites. In (Figure 3) these two binding sites are colored in magenta and red. Binding of at least one insulin to the red binding site in (Figure 3) is required for the activation of the insulin receptor and the change in conformation to the active T state. [3].
RelevanceAs mentioned in the Introduction, the insulin receptor is relevant to numerous biological functions of the body. In order to address the receptors role in glucose homeostasis, it is important to discuss its relevance in disease. In a healthy, normal-functioning human, each cell's insulin receptors have the ability to bind to an abundance of insulin that can get released by the pancreas in response to a rise of blood glucose levels. Without properly functioning insulin receptors that can respond to increases in insulin, and therefore glucose, medical intervention is necessary for survival. DiseaseOne of the most common diseases involving the insulin receptor in regards to glucose uptake and homeostasis is diabetes mellitus. There are two types of diabetes- which are referred to as type 1 and type 2 diabetes. Type 1 diabetes is classified as "insulin dependent" and is characterized by an inability for the body to produce insulin. This is most often the result of damage or insufficiency in the Islets of Langerhans in the pancreas. Type 2 diabetes is classified as "insulin independent" and is the result of the body producing insufficient amounts of insulin, or not responding to the insulin. This often occurs because of high blood-glucose levels. Both types of diabetes are often treated with insulin injections, and diet and lifestyle changes. [4] [5]. Treatment of Diabetes with Insulin At the Cellular LevelIn the human body, the conformation change from the inactive to active state upon insulin binding has a time constant of six minutes. Once insulin binds and the beta subunits are brought within close proximity, autophosphorylation of the beta subunits begins. Phosphorylation at these sites reaches a maximal level in about one minute, and lasts for approximately six to ten minutes. One insulin receptor substrate has a half-life of 3.5 minutes where it is able to be phosphorylated by the tyrosine kinases of the beta subunit and then act as a central hub to activate further downstream signaling pathways that eventually bring glucose receptors to the surface of the cell to allow for diffusion of glucose into the cell. Once insulin binds to the alpha subunit, the receptor remains active for approximately ten minutes before the insulin is degraded and the receptor returns to its inactive conformation. This time frame puts a perspective on how long it takes for the human body to store excess glucose in their blood stream from a recent meal as glycogen for later use as fuel.
An example for how to call up a reference in the future. You originally used this reference at the beginning of the Structural Overview. Now I have just taken the name in brackets and it will use the same number as the first time that this reference was used.
| ||||||||||||
References
- ↑ 1.0 1.1 Tatulian SA. Structural Dynamics of Insulin Receptor and Transmembrane Signaling. Biochemistry. 2015 Sep 15;54(36):5523-32. doi: 10.1021/acs.biochem.5b00805. Epub , 2015 Sep 3. PMID:26322622 doi:http://dx.doi.org/10.1021/acs.biochem.5b00805
- ↑ Weis F, Menting JG, Margetts MB, Chan SJ, Xu Y, Tennagels N, Wohlfart P, Langer T, Muller CW, Dreyer MK, Lawrence MC. The signalling conformation of the insulin receptor ectodomain. Nat Commun. 2018 Oct 24;9(1):4420. doi: 10.1038/s41467-018-06826-6. PMID:30356040 doi:http://dx.doi.org/10.1038/s41467-018-06826-6
- ↑ Uchikawa E, Choi E, Shang G, Yu H, Bai XC. Activation mechanism of the insulin receptor revealed by cryo-EM structure of the fully liganded receptor-ligand complex. Elife. 2019 Aug 22;8. pii: 48630. doi: 10.7554/eLife.48630. PMID:31436533 doi:http://dx.doi.org/10.7554/eLife.48630
- ↑ Wilcox G. Insulin and insulin resistance. Clin Biochem Rev. 2005 May;26(2):19-39. PMID:16278749
- ↑ Riddle MC. Treatment of diabetes with insulin. From art to science. West J Med. 1983 Jun;138(6):838-46. PMID:6351440
Student Contributors
- Harrison Smith
- Alyssa Ritter
