This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Journal:Proteins:3
From Proteopedia

Do Newly Born orphan proteins resemble Never Born proteins? A study using three deep learning algorithms
Newly Born proteins, or orphan proteins, have no sequence homology to other proteins and occur in single species or within a taxonomically restricted gene family
Never Born proteins are random polypeptides with amino acid content similar to that of native proteins.
Can recently developed AI/Deep Learning tools for predicting 3D protein structures like:
- AlphaFold2 (AF2)
- RoseTTAFold (RTF)
- Evolutionary Scale Modeling (ESM-2)
be useful to see if Newly Born proteins are similar to Never Born proteins? AF2 and RTF predict, by default, five top models, while ESM-2 predicts only one model. Morphing between the top models of AF2 and those of RTF give a visual feeling of how similar these 5 models are for each method.
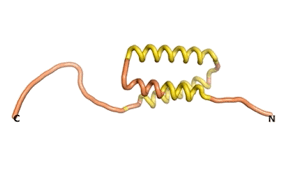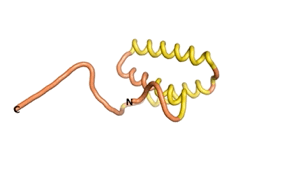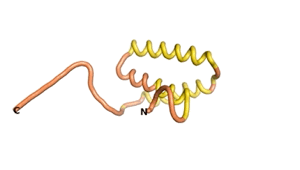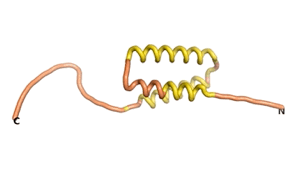
Sequences of the Never Born proteins by Tretyachenko et al.[1] showed that some folded into compact structures, e.g., as seen for Sequences #1856 and #6387.
| RTF-1856 | ESM-1856 | AF2-1856 |
|---|---|---|
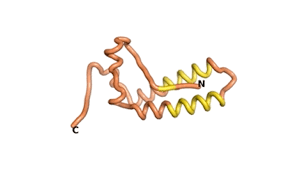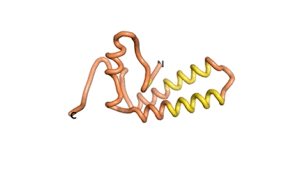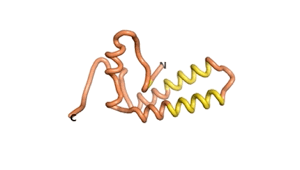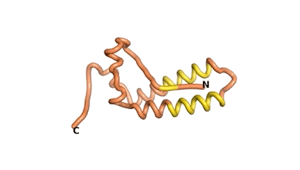
| 
| 
|
| RTF-6387 | ESM-6387 | AF2-6387 |
|---|---|---|

| 
| 
|
Other never born proteins experimentally appear to belong to the category of intrinsically disordered proteins (IDPs)[2], e.g., as seen for Sequence #3703.
| RTF-3703 | ESM-3703 | AF2-3703 |
|---|---|---|

| Image:ESM 35.jpg.jpg | 
|
References
- ↑ Tretyachenko V, Vymětal J, Bednárová L, Kopecký V Jr, Hofbauerová K, Jindrová H, Hubálek M, Souček R, Konvalinka J, Vondrášek J, Hlouchová K. Random protein sequences can form defined secondary structures and are well-tolerated in vivo. Sci Rep. 2017 Nov 13;7(1):15449. PMID:29133927 doi:10.1038/s41598-017-15635-8
- ↑ Dunker AK, Silman I, Uversky VN, Sussman JL. Function and structure of inherently disordered proteins. Curr Opin Struct Biol. 2008 Dec;18(6):756-64. Epub 2008 Nov 17. PMID:18952168 doi:10.1016/j.sbi.2008.10.002
sample
