Papain
From Proteopedia
Contents |
Introduction
Papain. Meat tenderizer. Old time home remedy for insect, jellyfish, and stingray stings[1]. Who would have thought that a sulfhydryl protease from the latex of the papaya fruit, Carica papaya and Vasconcellea cundinamarcensis would have such a practical application beyond proteopedia?
Introduction
General Characteristics
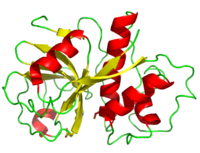
Papain is a 23.4 kDa, 212 residue cysteine protease, also known as papaya proteinase I, from the peptidase C1 family (E.C. 3.4.22.2).[2][3] It is the natural product of the Papaya(Carica papaya)[4], and may be extracted from the plant's latex, leaves and roots.[5] Papain displays a broad range of functions, acting as an endopeptidase, amidase, and esterase,[6] with its optimal activity values for pH lying between 6.0 and 7.0, and its optimal temperature for activity is 65 °C. Its pI values are 8.75 and 9.55, and it is best visualized at a wavelength of 278 nm.[7]
Common Uses
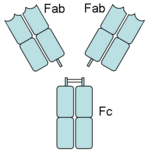
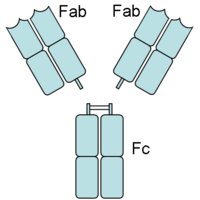
Papain digests most proteins, often more extensively than pancreatic proteases. It has a very broad specificity and is known to cleave peptide bonds of basic amino acids and leucine and glycine residues, but prefers amino acids with large hydrophobic side chains. This non-specific nature of papain's hydrolase activity has led to its use in many and varied commercial products. It is often used as a meat tenderizer because it can hydrolyze the peptide bonds of collagen, elastin, and actomyosin. It is also used in contact lens solution to remove protein deposits on the lenses and marketed as a digestive supplement. Another place papain has found use is clinical medicine, where it is used to treat pain, swelling, and fluid retention following trauma and surgery. [8] Finally, papain has several common uses in general biomedical research, including a gentle cell isolation agent, production of glycopeptides from purified proteoglycans, and solubilization of integral membrane proteins. It is also notable for its ability to specifically cleave IgG and IgM antibodies above and below the disulfide bonds that join the heavy chains and that is found between the light chain and heavy chain. This generates two monovalent Fab segments, that each have a single antibody binding sites, and an intact Fc fragment, as shown in the image to the right:[6]
History
Papain's enzymatic use was first discovered in 1873 by G.C. Roy who published his results in the Calcutta Medical Journal in the article, "The Solvent Action of Papaya Juice on Nitrogenous Articles of Food." In 1879, papain was named officially by Wurtz and Bouchut, who managed to partially purify the product from the sap of papaya. It wasn't until the mid-twentieth century that the complete purification and isolation of papain was achieved. In 1968, Drenth et al. determined the structure of papain by x-ray crystallography, making it the second enzyme whose structure was successfully determined by x-ray crystallography. Additionally, papain was the first cysteine protease to have its structure identified.[6] In 1984, Kamphuis et al. determined the geometry of the active site, and the three-dimensional structure was visualized to a 1.65 Angstrom solution.[9] Today, studies continue on the stability of papain, involving changes in environmental conditions as well as testing of inhibitors such as phenylmethanesulfonylfluoride (PMSF), TLCK, TPCK, aplh2-macroglobulin, heavy metals, AEBSF, antipain, cystatin, E-64, leupeptin, sulfhydryl binding agents, carbonyl reagents, and alkylating agents.[6]
Structure
|
General Structural Features
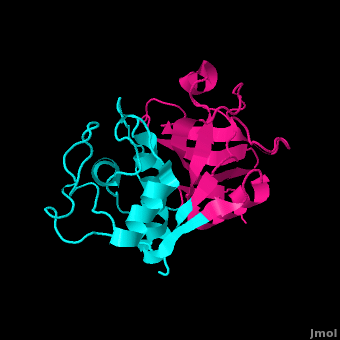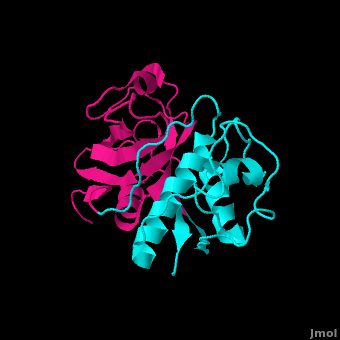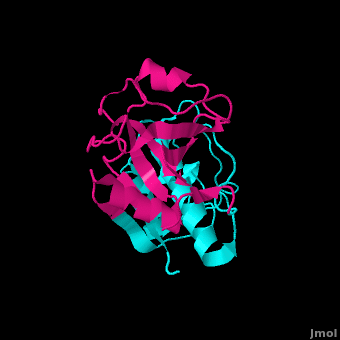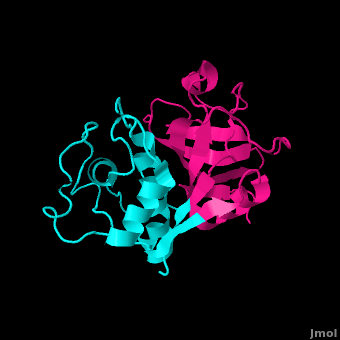
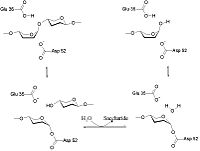
Papain is a relatively simple enzyme, consisting of a single 212 residue chain. A majority of papain's residues, shown in purple in the link, are . As with all proteins, it is primarily the exclusion of these residues by water that leads to papain's assumption of a globular form. Despite its apparent simplicity and small size, papain folds into two distinct, evenly sized , each with its own . These two subunits are held together with "arm" linkage, pictured to the left, where one end of the protein chain holds the opposite domain. In papain's case the "arm crossing" primarily occurs on or near the surface.[10] It is between these two domains that the is situated.[11] The two domains interact with one another via hydrophobic interactions, hydrogen bonds, and electrostatic interactions in this cleft. For example, from the L Domain hydrophobically interacts with the carbon atoms on residues Lys174, Ala162 and Pro129 of the R Domain. hydrogen bonds multiple times with the oxygen atoms of Ser176 and also with the oxygen atom on Tyr88. Electrostatic interactions are seen between where the carboxyl group of Glu35 forms an ionic bond with the ammonia group of the Lys174 residue. The sum total of interactions within the cleft between the two domains ensure that the lobes do not move with respect to one another. [12]
In addition to hydrophobic residues, papain contains a variety of residues, some carrying a (acidic) at physiological pH, others a (basic), the rest of the polar residues are neutral. As expected, the charged face outward due to their hydrophilic nature.
Papain's secondary structure is composed of 21% (45 residues comprising 17 sheets) and 25% (51 residues comprising 7 helices). The rest of the residues, accounting for over 50% of the enzymes structure, make up ordered non-repetative sequences.[13] These secondary structures may be traced from the N- to C-terminus by means of . As shown in this scene, the red end begins the protein at the N-terminus, and can be traced through the colors of the rainbow to the blue end at the C-terminus. These secondary structures form as a result of favorable hydrogen bonding interactions within the polypeptide backbone. Meanwhile, secondary structures are kept in place by hydrophobic interactions and hydrogen bonds between sidechains of adjacent structures. For example, the (residues 25-42) is maintained as a result of between backbone carbonyl atoms and the hydrogen on the amide nitrogen four residues away. However, are present between this helix and the rest of the protein, suggesting that this helix is coordinated primarily by hydrophobic interactions. This is reasonable given its central location in the enzyme. As expected, the helix contains many (red residues are hydrophilic).
strongly contribute to the tertiary structure of papain.[14] In this particular image, clarification of residue coordination is demonstrated by color: paired residues are shown in the same color, oxygen is shown in red, and nitrogen is shown in blue. The tertiary structure of papain is also maintained by three , which connect , , and [3]. These disulfide bonds are likely important in conserving the structural integrity of the enzyme as it operates in extracellular environments at high temperatures.
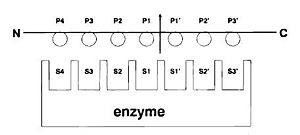
Active Site and Substrate Binding
Papain
Introduction: Did you know?
. Meat tenderizer. Old time home remedy for insect, jellyfish, and stingray stings[15]. Who would have thought that a sulfhydryl protease from the latex of the papaya fruit, Carica papaya and Vasconcellea cundinamarcensis would have such a practical application beyond proteopedia?
This protease belongs to an extended family of aminopeptidases, dipeptidyl peptidases, endopeptidases, and other enzymes having both exo- and endo-peptidase activity. The inactivated zymogen with N-terminal propeptide regions - providing stability in alkaline environments and enabling proper folding - is activated through removal of the propeptide regions. [16] The protein is primarily secreted with its pro-region enabling transport from zymogen to lysosome through membrane association and mediation. [17]
Papain made its first appearance in the Calcutta Medical Journal entitled “The Solvent Action of Papaya Juice on Nitrogenous Articles of Food” when G.C Roy was investigating the enzyme in 1873. In the late 19th century, Wurtz and Bouchut dubbed the partially purified enzyme "papain." [18] At the time, it was viewed as proteolytically active constituent in the latex of tropical papaya fruit. [19] As separation and purification techniques improved, pure papain was able to be isolated. In becoming the second enzyme to attain an X-ray crystallized structure and the first cysteine protease to behold an identifiable structure, papain fueled greater advances in enzymatic studies. [20]
Medicinal Uses
Papain has been used for a plethora of medicinal purposes including treating inflammation, shingles, diarrhea, psoriasis, parasites, and many others.[21] One major use is the treatment of cutaneous ulcers including diabetic ulcers and pressure ulcers.[22] Pressures ulcers plague many bed bound individuals and are a major source of pain and discomfort. Two papain based topical drugs are Accuzyme and Panafil, which can be used to treat wounds like cutaneous ulcers.[23]
Papain in The News

A recent New York Times article featured papain and other digestive enzymes.[25] With the number of individuals suffering from irritable bowel syndrome and other gastrointestinal issues, many people are turning toward natural digestive aid supplements like papain. The author even talks about the use of papain along with a pineapple enzyme, bromelain, in cosmetic facial masks. Dr. Adam R. Kolker (a plastic surgeon) is quoted in the article saying that "For skin that is sensitive, enzymes are wonderful." He bases these claims off the idea that proteases like papain help to break peptide bonds holding dead skin cells to the live skin cells.[26]
==Structure==
| |||||||||||
| |||||||||||
Proteopedia Page Contributors and Editors (what is this?)
Kirsten Eldredge, Jacinth Koh, Sara Kongkatong, Kyle Burch, Michal Harel, Joel L. Sussman, Elizabeth Miller, Samuel Bray, David Canner, Jaime Prilusky