Background
Transpeptidases (TP), also known as penicillin-binding proteins (PBP), catalyze the cross-linking of peptidoglycan polymers during bacterial cell wall synthesis. The natural transpeptidase substrate is the D-Ala-D-Ala peptidoglycan side chain terminus. Beta-lactam (β-lactam) antibiotics, which include penicillins, cephalosporins and carbapenems, bind and irreversibly inhibit transpeptidases by mimicking the D-Ala-D-Ala substrate, resulting in the inhibition of cell wall synthesis and ultimately bacterial cell growth.
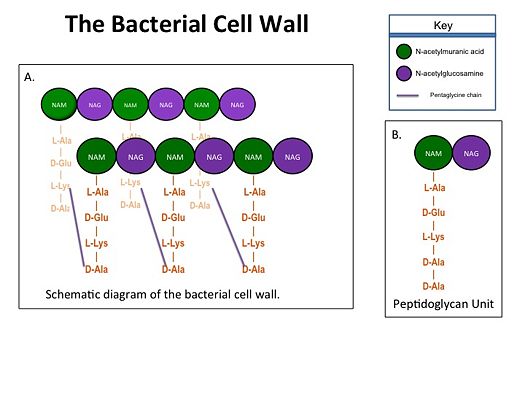
Figure 1. A.Bacterial Cell Wall B.Peptidoglycan with D-Ala-D-Ala substrate
Overuse and misuse of β-lactams has led to the generation of methicillin- resistant Staphylococcus aureus (MRSA) isolates that have acquired an alternative transpeptidase, PBP2a, which is neither bound nor inhibited by β- lactams. MRSA isolates are resistant to all β-lactams, and are often only susceptible to “last resort antibiotics”, such as vancomycin. Recently, two cephalosporins - ceftobiprole and ceftaroline - that bind and inhibit PBP2a have been developed.
How does PBP2a work