This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
4ald
From Proteopedia
|
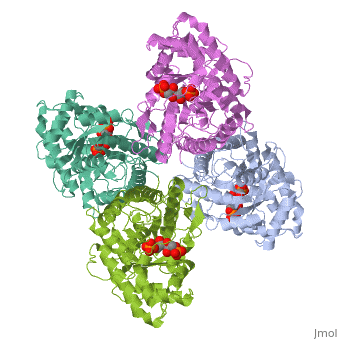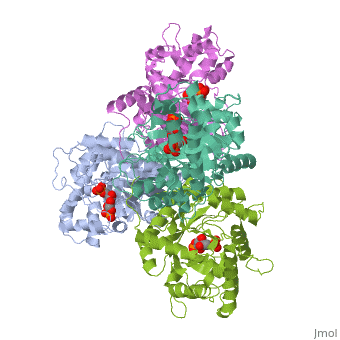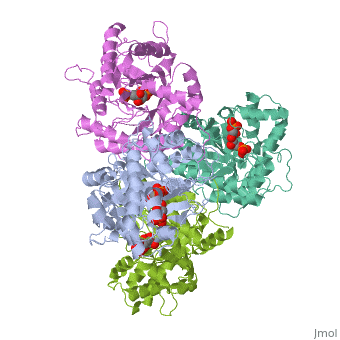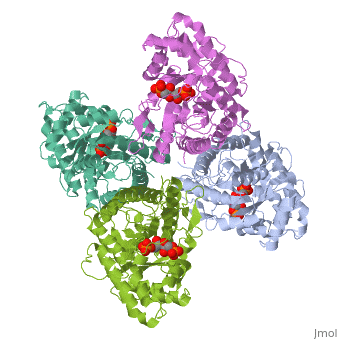
HUMAN MUSCLE FRUCTOSE 1,6-BISPHOSPHATE ALDOLASE COMPLEXED WITH FRUCTOSE 1,6-BISPHOSPHATE
Overview
Fructose 1,6-bisphosphate aldolase catalyzes the reversible cleavage of, fructose 1,6-bisphosphate and fructose 1-phosphate to dihydroxyacetone, phosphate and either glyceraldehyde 3-phosphate or glyceraldehyde, respectively. Catalysis involves the formation of a Schiff's base, intermediate formed at the epsilon-amino group of Lys229. The existing, apo-enzyme structure was refined using the crystallographic free-R-factor, and maximum likelihood methods that have been shown to give improved, structural results that are less subject to model bias. Crystals were also, soaked with the natural substrate (fructose 1,6-bisphosphate), and the, crystal structure of this complex has been determined to 2.8 A. The apo, structure differs from the previous Brookhaven-deposited structure (1ald), in the ... [(full description)]
About this Structure
4ALD is a [Single protein] structure of sequence from [Homo sapiens] with 2FP as [ligand]. The following page contains interesting information on the relation of 4ALD with [The Glycolytic Enzymes]. Active as [Fructose-bisphosphate aldolase], with EC number [4.1.2.13]. Structure known Active Site: SBL. Full crystallographic information is available from [OCA].
Reference
Crystal structure of human muscle aldolase complexed with fructose 1,6-bisphosphate: mechanistic implications., Dalby A, Dauter Z, Littlechild JA, Protein Sci. 1999 Feb;8(2):291-7. PMID:10048322
Page seeded by OCA on Tue Oct 30 17:47:20 2007