This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
3ep2
From Proteopedia
| |||||||||
| 3ep2, resolution 9.00Å () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Related: | 2avy, 2aw4, 1qza, 1ob2, 3eq3, 3eq4 | ||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, PDBsum, RCSB | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
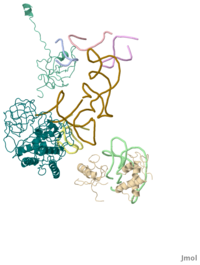
Model of Phe-tRNA(Phe) in the ribosomal pre-accommodated state revealed by cryo-EM
The accuracy of ribosomal translation is achieved by an initial selection and a proofreading step, mediated by EF-Tu, which forms a ternary complex with aminoacyl(aa)-tRNA. To study the binding modes of different aa-tRNAs, we compared cryo-EM maps of the kirromycin-stalled ribosome bound with ternary complexes containing Phe-tRNA(Phe), Trp-tRNA(Trp), or Leu-tRNA(LeuI). The three maps suggest a common binding manner of cognate aa-tRNAs in their specific binding with both the ribosome and EF-Tu. All three aa-tRNAs have the same 'loaded spring' conformation with a kink and twist between the D-stem and anticodon stem. The three complexes are similarly integrated in an interaction network, extending from the anticodon loop through h44 and protein S12 to the EF-Tu-binding CCA end of aa-tRNA, proposed to signal cognate codon-anticodon interaction to the GTPase centre and tune the accuracy of aa-tRNA selection.
Recognition of aminoacyl-tRNA: a common molecular mechanism revealed by cryo-EM., Li W, Agirrezabala X, Lei J, Bouakaz L, Brunelle JL, Ortiz-Meoz RF, Green R, Sanyal S, Ehrenberg M, Frank J, EMBO J. 2008 Dec 17;27(24):3322-31. Epub 2008 Nov 20. PMID:19020518
From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.
About this Structure
3EP2 is a 9 chains structure of sequences from Escherichia coli k12. Full crystallographic information is available from OCA.
Reference
Recognition of aminoacyl-tRNA: a common molecular mechanism revealed by cryo-EM., Li W, Agirrezabala X, Lei J, Bouakaz L, Brunelle JL, Ortiz-Meoz RF, Green R, Sanyal S, Ehrenberg M, Frank J, EMBO J. 2008 Nov 20. PMID:19020518
Page seeded by OCA on Wed Dec 31 08:58:03 2008
Categories: Escherichia coli k12 | Agirrezabala, X. | Frank, J. | Li, W. | A/t-trna | Acetylation | Antibiotic resistance | Automated data collection | Cytoplasm | Elongation factor | Gtp-binding | Membrane | Methylation | Nucleotide-binding | Phosphoprotein | Protein biosynthesis | Protein translation | Ribonucleoprotein | Ribosomal protein | Ribosomal protein/rna complex | Rna-binding | Rrna-binding | Ternary complex | Trna-binding