This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Nathan Line sandbox 1
From Proteopedia
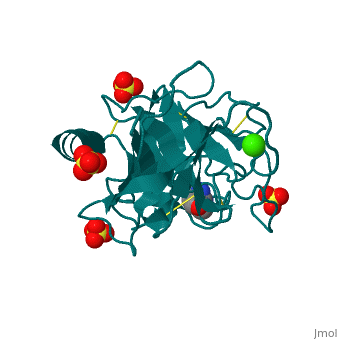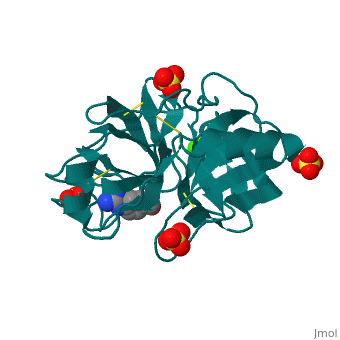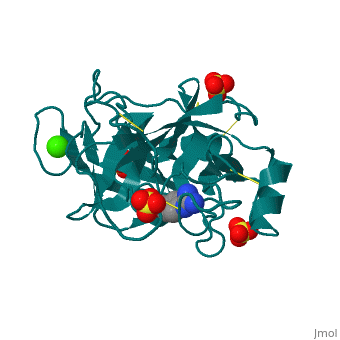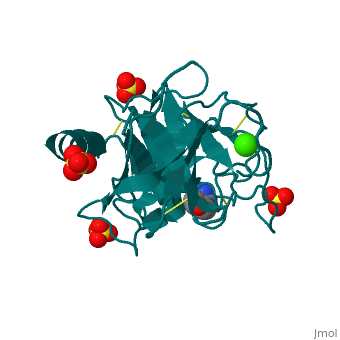
The Mechanism of Trypsin
Trypsin is a serine protease that breaks down the backbone of peptides near positively charged side chains. 2ah4 The peptide is pulled into the active site by Asp 189. His 57 pulls on the hydrogen attached to the OH on Ser 195 to make it a better nucleophilie which attacks the carbonyl of the peptide. This tetrahedral intermediate removes the hydrogen from His 57 while the carbonyl reforms resulting in a break in the peptide backbone. After the amino group is replaced with water, the water again attacks the carbonyl and this tetrahedral collapses to reconstruct both the Ser195 and His57. The final products result in an amino group and a carboxylic acid compound.
| |||||||||
| 2ah4, resolution 1.13Å () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ligands: | , , | ||||||||
| Activity: | Trypsin, with EC number 3.4.21.4 | ||||||||
| Related: | 2age, 2agg, 2agi | ||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, PDBsum, RCSB | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||