Sandbox 156
From Proteopedia
Contents |
Chloramphenicol Acetyltransferase Type III
Chloramphenicol acetyltransferase type III (CAT III) is an enzyme which catalyzes the transfer of the acetyl group from acetyl-CoA to hydroxyl groups of chloramphenicol. CAT III is a trimeric protein with a Mr of 25 000-kDa and a member of the actetyltransferase family of proteins.
Introduction
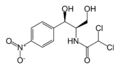
Found in bacteria, the CAT III enzyme is responsible for conferring resistance of the antibiotic chloramphenicol to the cell. Chloramphenicol arrests protein synthesis by binding to the bacterial ribosomes and causing the inhibition of peptidyl transferase activity[1]. However, when CAT III catalyzes the acetylation of chloramphenicol, the antibiotic can no longer bind to the ribosomes and is rendered inactive. The genes for the enzyme are commonly found on the plasmid of the bacteria and have been found in a numerous bacterial species[2].
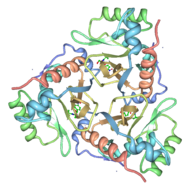
The multifunctional enzyme consists of three identical subunits with three active sites at the subunit interfaces. The side chains of one subunit allow van der Waals interactions and two hydrogen bonds with chloramphenicol, causing binding of the substrate. The opposing subunit provides a histidine (His-195) residue essential for catalysis[3]. Water molecules in the cavity provide a bridging hydrogen bond between the 1-hydroxyl of chloramphenicol and the hydroxyl of a threonine (Thr-174) residue. The active site of CAT III performs two acetylations of chloramphenicol and can accommodate the presence of the first intermediates quite well[4].
Reaction of CAT III
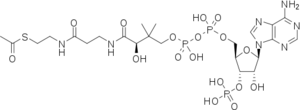
Both reactions take place in the active site of CAT III, where acetyl-CoA is tunneled through from the opposing side of the trimer[5].
In the first step of the reaction, Histidine-195 abstracts a proton from the 3-hydroxyl of chloramphenicol, promoting a nucleophilic attack from the oxyanion to the thioester bond of the acetyl-CoA. The intermediate produced, 3-acetylchloramphenicol, then rearranges non-enzymatically to 1-acetylchloramphenicol. Regeneration of the 3-hydroxyl allows another round of CAT III catalyzed nucleophilic attack and a 1,3-diacetylchloramphenicol product is formed[6].
Structure
|
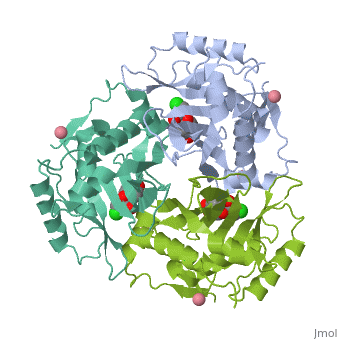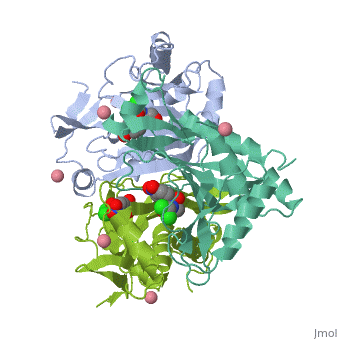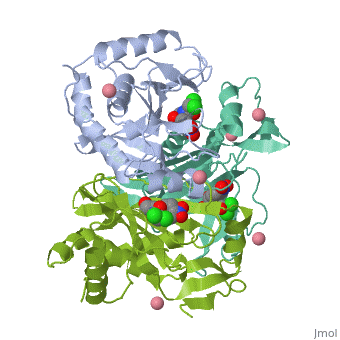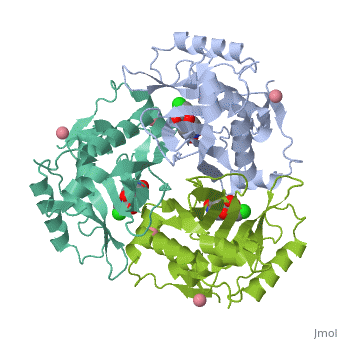
The general structure of CAT III is dominated by a six stranded antiparallel and 5 stacked against the ends and face of the protein, forming a structure known as an "open-faced sandwhich"[7]. An extended β-strand forms an extension of the six stranded sheet to a seven stranded sheet that spans the interface of the subunit. Three identical monomers associate to form the trimeric protein with two ions acting as cofactors.
The Chloramphenicol Binding Site
The association of the monomeric subunits produces a well-defined pocket at the subunit interface, allowing for the binding of the chloramphenicol molecule. The of CAT III is lined with hydrophobic residues, allowing